User:Mr. Ibrahem/Lamotrigine
| |||
| Clinical data | |||
|---|---|---|---|
| Pronunciation | /ləˈmoʊtrɪˌdʒiːn/ | ||
| Trade names | Lamictal, others[1] | ||
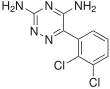
| Other names | BW-430C; BW430C; 3,5-Diamino-6-(2,3-dichlorophenyl)-1,2,4-triazine | ||
| AHFS/Drugs.com | Monograph | ||
| MedlinePlus | a695007 | ||
| License data |
| ||
| Pregnancy category |
| ||
| Routes of administration | By mouth | ||
| Drug class | Anticonvulsant[3] | ||
| Legal status | |||
| Legal status | |||
| Pharmacokinetic data | |||
| Bioavailability | 98% | ||
| Protein binding | 55% | ||
| Metabolism | Liver (mostly UGT1A4-mediated) | ||
| Elimination half-life | 29 hours | ||
| Excretion | Urine (65%), feces (2%) | ||
| Identifiers | |||
| |||
| Chemical and physical data | |||
| Formula | C9H7Cl2N5 | ||
| Molar mass | 256.091 g/mol | ||
| 3D model (JSmol) | |||
| |||
| |||
| | |||
Lamotrigine, sold as the brand name Lamictal among others, is an anticonvulsant medication used to treat epilepsy and bipolar disorder.[3] For epilepsy, this includes focal seizures, tonic-clonic seizures, and seizures in Lennox-Gastaut syndrome.[3] In bipolar disorder, it is used to treat acute episodes of depression and rapid cycling in bipolar type II and to prevent recurrence in bipolar type I.[3]
Common side effects include sleepiness, headache, vomiting, trouble with coordination, and rash.[3] Serious side effects include lack of red blood cells, increased risk of suicide, Stevens–Johnson syndrome, and allergic reactions.[3] Concerns exist that use during pregnancy or breastfeeding may result in harm.[2] Lamotrigine is a phenyltriazine, making it chemically different from other anticonvulsants.[3] How it works is not exactly clear.[3] It appears to decrease voltage-sensitive sodium channels of neurons.[3][4]
Lamotrigine was first marketed in the United Kingdom in 1991, and approved for use in the United States in 1994.[3][5] It is on the World Health Organization's List of Essential Medicines.[6] The wholesale cost in the developing world is about US$3.50 to US$23 per month as of 2015.[7] In the United States, this amount has a wholesale cost of about US$4.60 as of 2019.[8] In 2017, it was the 63rd most commonly prescribed medication in the United States, with more than twelve million prescriptions.[9][10]
References[edit]
- ^ Cite error: The named reference
brandswas invoked but never defined (see the help page). - ^ a b c "Lamotrigine Use During Pregnancy". Drugs.com. 8 October 2019. Archived from the original on 25 January 2021. Retrieved 24 March 2020.
- ^ a b c d e f g h i j k l "Lamotrigine". The American Society of Health-System Pharmacists. Archived from the original on 10 December 2017. Retrieved 8 December 2017.
- ^ "Lamotrigine". PubChem Open Chemistry Database. US: National Institutes of Health. Archived from the original on September 6, 2016. Retrieved December 13, 2016.
- ^ Shorvon SD, Perucca E, Engel J (2015). The Treatment of Epilepsy (4th ed.). John Wiley & Sons, Incorporated. p. 1321. ISBN 9781118936993. Archived from the original on 2017-12-10. Retrieved 2017-12-09.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Single Drug Information". International Medical Products Price Guide. Archived from the original on 10 December 2017. Retrieved 9 December 2017.
- ^ "NADAC as of 2019-11-27". Centers for Medicare and Medicaid Services. Archived from the original on 2020-07-11. Retrieved 26 November 2019.
- ^ "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ^ "Lamotrigine - Drug Usage Statistics". ClinCalc. 23 December 2019. Archived from the original on 28 February 2020. Retrieved 11 April 2020.