User:InterstellarGamer12321/Silicon compounds
| X = | C | Si | H | F | Cl | Br | I | O– | N< |
|---|---|---|---|---|---|---|---|---|---|
| C–X | 368 | 360 | 435 | 453 | 351 | 293 | 216 | ~360 | ~305 |
| Si–X | 360 | 340 | 393 | 565 | 381 | 310 | 234 | 452 | 322 |

Silicon compounds are compounds formed by the element silicon (Si). It mainly forms compounds in the -4 and +4 oxidation states, it can also form compounds in the −3, −2, −1, 0,[2] +1,[3] +2 and +3 oxidation states.
Crystalline bulk silicon is rather inert, but becomes more reactive at high temperatures. Like its neighbour aluminium, silicon forms a thin, continuous surface layer of silicon dioxide (SiO2) that protects the metal from oxidation. Thus silicon does not measurably react with the air below 900 °C, but formation of the vitreous dioxide rapidly increases between 950 °C and 1160 °C and when 1400 °C is reached, atmospheric nitrogen also reacts to give the nitrides SiN and Si3N4. Silicon reacts with gaseous sulfur at 600 °C and gaseous phosphorus at 1000 °C. This oxide layer nevertheless does not prevent reaction with the halogens; fluorine attacks silicon vigorously at room temperature, chlorine does so at about 300 °C, and bromine and iodine at about 500 °C. Silicon does not react with most aqueous acids, but is oxidised and complexed by hydrofluoric acid mixtures containing either chlorine or nitric acid to form hexafluorosilicates. It readily dissolves in hot aqueous alkali to form silicates.[4]
At high temperatures, silicon also reacts with alkyl halides; this reaction may be catalysed by copper to directly synthesise organosilicon chlorides as precursors to silicone polymers. Upon melting, silicon becomes extremely reactive, alloying with most metals to form silicides, and reducing most metal oxides because the heat of formation of silicon dioxide is so large. In fact, molten silicon reacts virtually with every known kind of crucible material (except its own oxide, SiO2).[5]: 13 This happens due to silicon's high binding forces for the light elements and to its high dissolving power for most elements.[5]: 13 As a result, containers for liquid silicon must be made of refractory, unreactive materials such as zirconium dioxide or group 4, 5, and 6 borides.[6][7]
Tetrahedral coordination is a major structural motif in silicon chemistry just as it is for carbon chemistry. However, the 3p subshell is rather more diffuse than the 2p subshell and does not hybridise so well with the 3s subshell. As a result, the chemistry of silicon and its heavier congeners shows significant differences from that of carbon,[8] and thus octahedral coordination is also significant.[6] For example, the electronegativity of silicon (1.90) is much less than that of carbon (2.55), because the valence electrons of silicon are further from the nucleus than those of carbon and hence experience smaller electrostatic forces of attraction from the nucleus. The poor overlap of 3p orbitals also results in a much lower tendency toward catenation (formation of Si–Si bonds) for silicon than for carbon, due to the concomitant weakening of the Si–Si bond compared to the C–C bond:[9] the average Si–Si bond energy is approximately 226 kJ/mol, compared to a value of 356 kJ/mol for the C–C bond.[10] This results in multiply bonded silicon compounds generally being much less stable than their carbon counterparts, an example of the double bond rule. On the other hand, the presence of radial nodes in the 3p orbitals of silicon suggests the possibility of hypervalence, as seen in five and six-coordinate derivatives of silicon such as SiX−
5 and SiF2−
6.[11][9] Lastly, because of the increasing energy gap between the valence s and p orbitals as the group is descended, the divalent state grows in importance from carbon to lead, so that a few unstable divalent compounds are known for silicon; this lowering of the main oxidation state, in tandem with increasing atomic radii, results in an increase of metallic character down the group. Silicon already shows some incipient metallic behavior, particularly in the behavior of its oxide compounds and its reaction with acids as well as bases (though this takes some effort), and is hence often referred to as a metalloid rather than a nonmetal.[9] However, metallicity does not become clear in group 14 until germanium and dominant until tin, with the growing importance of the lower +2 oxidation state.[12]
Silicides
[edit]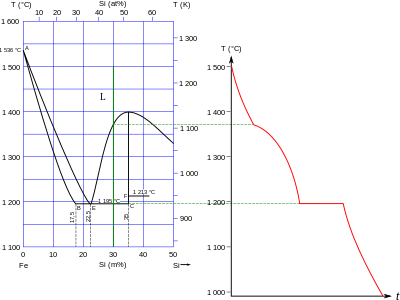
Many metal silicides are known, most of which have formulae that cannot be explained through simple appeals to valence: their bonding ranges from metallic to ionic and covalent. Some known stoichiometries are M6Si, M5Si, M4Si, M15Si4, M3Si, M5Si2, M2Si, M5Si3, M3Si2, MSi, M2Si3, MSi2, MSi3, and MSi6. They are structurally more similar to the borides than the carbides, in keeping with the diagonal relationship between boron and silicon, although the larger size of silicon than boron means that exact structural analogies are few and far between. The heats of formation of the silicides are usually similar to those of the borides and carbides of the same elements, but they usually melt at lower temperatures.[13] Silicides are known for all stable elements in groups 1–10, with the exception of beryllium: in particular, uranium and the transition metals of groups 4–10 show the widest range of stoichiometries. Except for copper, the metals in groups 11–15 do not form silicides. Instead, most form eutectic mixtures, although the heaviest post-transition metals mercury, thallium, lead, and bismuth are completely immiscible with liquid silicon.[6]
Usually, silicides are prepared by direct reaction of the elements. For example, the alkali metals and alkaline earth metals react with silicon or silicon oxide to give silicides. Nevertheless, even with these highly electropositive elements true silicon anions are not obtainable, and most of these compounds are semiconductors. For example, the alkali metal silicides (M+
)
4(Si4−
4) contain pyramidal tricoordinate silicon in the Si4−
4 anion, isoelectronic with white phosphorus, P4.[6][14] Metal-rich silicides tend to have isolated silicon atoms (e. g. Cu5Si); with increasing silicon content, catenation increases, resulting in isolated clusters of two (e. g. U3Si2) or four silicon atoms (e. g. [K+]4[Si4]4−) at first, followed by chains (e. g. CaSi), layers (e. g. CaSi2), or three-dimensional networks of silicon atoms spanning space (e. g. α-ThSi2) as the silicon content rises even higher.[6]
The silicides of the group 1 and 2 metals usually are more reactive than the transition metal silicides. The latter usually do not react with aqueous reagents, except for hydrofluoric acid; however, they do react with much more aggressive reagents such as liquid potassium hydroxide, or gaseous fluorine or chlorine when red-hot. The pre-transition metal silicides instead readily react with water and aqueous acids, usually producing hydrogen or silanes:[6]
- Na2Si + 3 H2O → Na2SiO3 + 3 H2
- Mg2Si + 2 H2SO4 → 2 MgSO4 + SiH4
Products often vary with the stoichiometry of the silicide reactant. For example, Ca2Si is polar and non-conducting and has the anti-PbCl2 structure with single isolated silicon atoms, and reacts with water to produce calcium hydroxide, hydrated silicon dioxide, and hydrogen gas. CaSi with its zigzag chains of silicon atoms instead reacts to give silanes and polymeric SiH2, while CaSi2 with its puckered layers of silicon atoms does not react with water, but will react with dilute hydrochloric acid: the product is a yellow polymeric solid with stoichiometry Si2H2O.[6]
Silanes
[edit]Speculation on silicon hydride chemistry started in the 1830s, contemporary with the development of synthetic organic chemistry. Silane itself, as well as trichlorosilane, were first synthesised by Friedrich Wöhler and Heinrich Buff in 1857 by reacting aluminium–silicon alloys with hydrochloric acid, and characterised as SiH4 and SiHCl3 by Charles Friedel and Albert Ladenburg in 1867. Disilane (Si2H6) followed in 1902, when it was first made by Henri Moissan and Samuel Smiles by the protonolysis of magnesium silicides. Further investigation had to wait until 1916 because of the great reactivity and thermal instability of the silanes; it was then that Alfred Stock began to study silicon hydrides in earnest with new greaseless vacuum techniques, as they were found as contaminants of his focus, the boron hydrides. The names silanes and boranes are his, based on analogy with the alkanes.[1][15][16] The Moissan and Smiles method of preparation of silanes and silane derivatives via protonolysis of metal silicides is still used, although the yield is lowered by the hydrolysis of the products that occurs simultaneously, so that the preferred route today is to treat substituted silanes with hydride reducing agents such as lithium aluminium hydride in etheric solutions at low temperatures. Direct reaction of HX or RX with silicon, possibly with a catalyst such as copper, is also a viable method of producing substituted silanes.[1]
The silanes comprise a homologous series of silicon hydrides with a general formula of SinH2n + 2. They are all strong reducing agents. Unbranched and branched chains are known up to n=8, and the cycles Si5H10 and Si6H12 are also known. The first two, silane and disilane, are colourless gases; the heavier members of the series are volatile liquids. All silanes are very reactive and catch fire or explode spontaneously in air. They become less thermally stable with room temperature, so that only silane is indefinitely stable at room temperature, although disilane does not decompose very quickly (only 2.5% of a sample decomposes after the passage of eight months).[1] They decompose to form polymeric polysilicon hydride and hydrogen gas.[17][18] As expected from the difference in atomic weight, the silanes are less volatile than the corresponding alkanes and boranes, but more so than the corresponding germanes. They are much more reactive than the corresponding alkanes, because of the larger radius of silicon compared to carbon facilitating nucleophilic attack at the silicon, the greater polarity of the Si–H bond compared to the C–H bond, and the ability of silicon to expand its octet and hence form adducts and lower the reaction's activation energy.[1]
Silane pyrolysis gives polymeric species and finally elemental silicon and hydrogen; indeed ultrapure silicon is commercially produced by the pyrolysis of silane. While the thermal decomposition of alkanes starts by the breaking of a C–H or C–C bond and the formation of radical intermediates, polysilanes decompose by eliminating silylenes :SiH2 or :SiHR, as the activation energy of this process (~210 kJ/mol) is much less than the Si–Si and Si–H bond energies. While pure silanes do not react with pure water or dilute acids, traces of alkali catalyse immediate hydrolysis to hydrated silicon dioxide. If the reaction is carried out in methanol, controlled solvolysis results in the products SiH2(OMe)2, SiH(OMe)3, and Si(OMe)4. The Si–H bond also adds to alkenes, a reaction which proceeds slowly and speeds up with increasing substitution of the silane involved. At 450 °C, silane participates in an addition reaction with acetone, as well as a ring-opening reaction with ethylene oxide. Direct reaction of the silanes with chlorine or bromine results in explosions at room temperature, but the reaction of silane with bromine at −80 °C is controlled and yields bromosilane and dibromosilane. The monohalosilanes may be formed by reacting silane with the appropriate hydrogen halide with an Al2X6 catalyst, or by reacting silane with a solid silver halide in a heated flow reactor:[1]
- SiH4 + 2 AgCl SiH3Cl + HCl + 2 Ag
Among the derivatives of silane, iodosilane (SiH3I) and potassium silanide (KSiH3) are very useful synthetic intermediates in the production of more complicated silicon-containing compounds: the latter is a colourless crystalline ionic solid containing K+ cations and SiH−
3 anions in the NaCl structure, and is made by the reduction of silane by potassium metal.[19] Additionally, the reactive hypervalent species SiH−
5 is also known.[1] With suitable organic substituents it is possible to produce stable polysilanes: they have surprisingly high electric conductivities, arising from sigma delocalisation of the electrons in the chain.[20]
Halides
[edit]Silicon and silicon carbide readily react with all four stable halogens, forming the colourless, reactive, and volatile silicon tetrahalides[21] Silicon tetrafluoride also may be made by fluorinating the other silicon halides, and is produced by the attack of hydrofluoric acid on glass.[22] Heating two different tetrahalides together also produces a random mixture of mixed halides, which may also be produced by halogen exchange reactions. The melting and boiling points of these species usually rise with increasing atomic weight, though there are many exceptions: for example, the melting and boiling points drop as one passes from SiFBr3 through SiFClBr2 to SiFCl2Br. The shift from the hypoelectronic elements in Group 13 and earlier to the Group 14 elements is illustrated by the change from an infinite ionic structure in aluminium fluoride to a lattice of simple covalent silicon tetrafluoride molecules, as dictated by the lower electronegativity of aluminium than silicon, the stoichiometry (the +4 oxidation state being too high for true ionicity), and the smaller size of the silicon atom compared to the aluminium atom.[21]
Silicon tetrachloride is manufactured on a huge scale as a precursor to the production of pure silicon, silicon dioxide, and some silicon esters.[21] The silicon tetrahalides hydrolyse readily in water, unlike the carbon tetrahalides, again because of the larger size of the silicon atom rendering it more open to nucleophilic attack and the ability of the silicon atom to expand its octet which carbon lacks.[22] The reaction of silicon tetrafluoride with excess hydrofluoric acid produces the octahedral hexafluorosilicate anion SiF2−
6.[22]
Analogous to the silanes, halopolysilanes SinX2n + 2 also are known. While catenation in carbon compounds is maximised in the hydrogen compounds rather than the halides, the opposite is true for silicon, so that the halopolysilanes are known up to at least Si14F30, Si6Cl14, and Si4Br10. A suggested explanation for this phenomenon is the compensation for the electron loss of silicon to the more electronegative halogen atoms by pi backbonding from the filled pπ orbitals on the halogen atoms to the empty dπ orbitals on silicon: this is similar to the situation of carbon monoxide in metal carbonyl complexes and explains their stability. These halopolysilanes may be produced by comproportionation of silicon tetrahalides with elemental silicon, or by condensation of lighter halopolysilanes (trimethylammonium being a useful catalyst for this reaction).[21]
Silica
[edit]Silicon dioxide (SiO2), also known as silica, is one of the best-studied compounds, second only to water. Twelve different crystal modifications of silica are known, the most common being α-quartz, a major constituent of many rocks such as granite and sandstone. It also is known to occur in a pure form as rock crystal; impure forms are known as rose quartz, smoky quartz, morion, amethyst, and citrine. Some poorly crystalline forms of quartz are also known, such as chalcedony, chrysoprase, carnelian, agate, onyx, jasper, heliotrope, and flint. Other modifications of silicon dioxide are known in some other minerals such as tridymite and cristobalite, as well as the much less common coesite and stishovite. Biologically generated forms are also known as kieselguhr and diatomaceous earth. Vitreous silicon dioxide is known as tektites, and obsidian, and rarely as lechatelierite. Some synthetic forms are known as keatite. Opals are composed of complicated crystalline aggregates of partially hydrated silicon dioxide.[23]
-
Quartz
-
Agate
-
Tridymite
-
Cristobalite
-
Coesite
Most crystalline forms of silica are made of infinite arrangements of {SiO4} tetrahedra (with Si at the center) connected at their corners, with each oxygen atom linked to two silicon atoms. In the thermodynamically stable room-temperature form, α-quartz, these tetrahedra are linked in intertwined helical chains with two different Si–O distances (159.7 and 161.7 pm) with a Si–O–Si angle of 144°. These helices can be either left- or right-handed, so that individual α-quartz crystals are optically active. At 537 °C, this transforms quickly and reversibly into the similar β-quartz, with a change of the Si–O–Si angle to 155° but a retention of handedness. Further heating to 867 °C results in another reversible phase transition to β-tridymite, in which some Si–O bonds are broken to allow for the arrangement of the {SiO4} tetrahedra into a more open and less dense hexagonal structure. This transition is slow and hence tridymite occurs as a metastable mineral even below this transition temperature; when cooled to about 120 °C it quickly and reversibly transforms by slight displacements of individual silicon and oxygen atoms to α-tridymite, similarly to the transition from α-quartz to β-quartz. β-tridymite slowly transforms to cubic β-cristobalite at about 1470 °C, which once again exists metastably below this transition temperature and transforms at 200–280 °C to α-cristobalite via small atomic displacements. β-cristobalite melts at 1713 °C; the freezing of silica from the melt is quite slow and vitrification, or the formation of a glass, is likely to occur instead. In vitreous silica, the {SiO4} tetrahedra remain corner-connected, but the symmetry and periodicity of the crystalline forms are lost. Because of the slow conversions between these three forms, it is possible upon rapid heating to melt β-quartz (1550 °C) or β-tridymite (1703 °C). Silica boils at approximately 2800 °C. Other high-pressure forms of silica are known, such as coesite and stishovite: these are known in nature, formed under the shock pressure of a meteorite impact and then rapidly quenched to preserve the crystal structure. Similar melting and cooling of silica occurs following lightning strikes, forming glassy lechatelierite. W-silica is an unstable low-density form involving {SiO4} tetrahedra sharing opposite edges instead of corners, forming parallel chains similarly to silicon disulfide (SiS2) and silicon diselenide (SiSe2): it quickly returns to forming amorphous silica with heat or traces of water[24]

Silica is rather inert chemically. It is not attacked by any acids other than hydrofluoric acid. However, it slowly dissolves in hot concentrated alkalis, and does so rather quickly in fused metal hydroxides or carbonates, to give metal silicates. Among the elements, it is attacked only by fluorine at room temperature to form silicon tetrafluoride: hydrogen and carbon also react, but require temperatures over 1000 °C to do so. Silica nevertheless reacts with many metal and metalloid oxides to form a wide variety of compounds important in the glass and ceramic industries above all, but also have many other uses: for example, sodium silicate is often used in detergents due to its buffering, saponifying, and emulsifying properties[24]
Silicic acids
[edit]Adding water to silica drops its melting point by around 800 °C due to the breaking of the structure by replacing Si–O–Si linkages with terminating Si–OH groups. Increasing water concentration results in the formation of hydrated silica gels and colloidal silica dispersions. Many hydrates and silicic acids exist in the most dilute of aqueous solutions, but these are rather insoluble and quickly precipitate and condense and cross-link to form various polysilicic acids of variable combinations following the formula [SiOx(OH)4−2x]n, similar to the behaviour of boron, aluminium, and iron, among other elements. Hence, although some simple silicic acids have been identified in dilute solutions, such as orthosilicic acid Si(OH)4 and metasilicic acid SiO(OH)2, none of these are likely to exist in the solid state.[24]
Silicate minerals
[edit]| CN 4 | LiI (59) |
BeII (27) | AlIII (39) | SiIV (26) | |
|---|---|---|---|---|---|
| CN 6 | NaI (102) | MgII (72) | AlIII (54) | TiIV (61) | FeII (78) |
| CN 8 | KI (151) | CaII (112) | |||
| CN 12 | KI (164) |
About 95% of the Earth's crustal rocks are made of silica or silicate and aluminosilicate minerals, as reflected in oxygen, silicon, and aluminium being the three most common elements in the crust (in that order).[25] Measured by mass, silicon makes up 27.7% of the Earth's crust.[26] Pure silicon crystals are very rarely found in nature, but notable exceptions are crystals as large as to 0.3 mm across found during sampling gases from the Kudriavy volcano on Iturup, one of the Kuril Islands.[27][28]
Silicate and aluminosilicate minerals have many different structures and varying stoichiometry, but they may be classified following some general principles. Tetrahedral {SiO4} units are common to almost all these compounds, either as discrete structures, or combined into larger units by the sharing of corner oxygen atoms. These may be divided into neso-silicates (discrete {SiO4} units) sharing no oxygen atoms, soro-silicates (discrete {Si2O7} units) sharing one, cyclo-silicates (closed ring structures) and ino-silicates (continuous chain or ribbon structures) both sharing two, phyllo-silicates (continuous sheets) sharing three, and tecto-silicates (continuous three-dimensional frameworks) sharing four. The lattice of oxygen atoms that results is usually close-packed, or close to it, with the charge being balanced by other cations in various different polyhedral sites according to size.[29]
The orthosilicates MII
2SiO
4 (M = Be, Mg, Mn, Fe, Zn) and ZrSiO4 are neso-silicates. Be
2SiO
4 (phenacite) is unusual as both BeII and SiIV occupy tetrahedral four-coordinated sites; the other divalent cations instead occupy six-coordinated octahedral sites and often isomorphously replace each other as in olivine, (Mg,Fe,Mn)2SiO4. Zircon, ZrSiO4, demands eight-coordination of the ZrIV cations due to stoichiometry and because of their larger ionic radius (84 pm). Also significant are the garnets, [MII
3MIII
2(SiO
4)
3], in which the divalent cations (e.g. Ca, Mg, Fe) are eight-coordinated and the trivalent ones are six-coordinated (e.g. Al, Cr, Fe). Regular coordination is not always present: for example, it is not found in Ca2SiO4, which mixes six- and eight-coordinate sites for CaII. Soro-silicates, involving discrete double or triple tetrahedral units, are quite rare: metasilicates involving cyclic "[(SiO3)n]2n−" units of corner-abutting tetrahedra forming a polygonal ring are also known.[25]
Chain metasilicates, {SiO2−
3}
∞, form by corner-sharing of an indefinite chain of linked {SiO4} tetrahedra. Many differences arise due to the differing repeat distances of conformation across the line of tetrahedra. A repeat distance of two is most common, as in most pyroxene minerals, but repeat distances of one, three, four, five, six, seven, nine, and twelve are also known. These chains may then link across each other to form double chains and ribbons, as in the asbestos minerals, involving repeated chains of cyclic tetrahedron rings.[25]

Layer silicates, such as the clay minerals and the micas, are very common, and often are formed by horizontal cross-linking of metasilicate chains or planar condensation of smaller units. An example is kaolinite [Al2(OH)4Si2O5]; in many of these minerals cation and anion replacement is common, so that for example tetrahedral SiIV may be replaced by AlIII, octahedral AlIII by MgII, and OH− by F−. Three-dimensional framework aluminosilicates are structurally very complex; they may be conceived of as starting from the SiO2 structure, but having replaced up to one-half of the SiIV atoms with AlIII, they require more cations to be included in the structure to balance charge. Examples include feldspars (the most abundant minerals on the Earth), zeolites, and ultramarines. Many feldspars can be thought of as forming part of the ternary system NaAlSi3O8–KAlSi3O8–CaAl2Si2O8. Their lattice is destroyed by high pressure prompting AlIII to undergo six-coordination rather than four-coordination, and this reaction destroying feldspars may be a reason for the Mohorovičić discontinuity, which would imply that the crust and mantle have the same chemical composition, but different lattices, although this is not a universally held view. Zeolites have many polyhedral cavities in their frameworks (truncated cuboctahedra being most common, but other polyhedra also are known as zeolite cavities), allowing them to include loosely bound molecules such as water in their structure. Ultramarines alternate silicon and aluminium atoms and include a variety of other anions such as Cl−, SO2−
4, and S2−
2, but are otherwise similar to the feldspars.[25]
Other inorganic compounds
[edit]Silicon disulfide (SiS2) is formed by burning silicon in gaseous sulfur at 100 °C; sublimation of the resulting compound in nitrogen results in white, flexible long fibers reminiscent of asbestos with a structure similar to W-silica. This melts at 1090 °C and sublimes at 1250 °C; at high temperature and pressure this transforms to a crystal structure analogous to cristobalite. However, SiS2 lacks the variety of structures of SiO2, and quickly hydrolyses to silica and hydrogen sulfide. It is also ammonolysed quickly and completely by liquid ammonia as follows to form an imide:[30]
- SiS2 + 4 NH3 → Si(NH)2 + 2 NH4SH
It reacts with the sulfides of sodium, magnesium, aluminium, and iron to form metal thiosilicates: reaction with ethanol results in tetraethylsilicate Si(OEt)4 and hydrogen sulfide. Ethylsilicate is useful as its controlled hydrolysis produces adhesive or film-like forms of silica. Reacting hydrogen sulfide with silicon tetrahalides yields silicon thiohalides such as S(SiCl)3, cyclic Cl2Si(μ-S)2SiCl2, and crystalline (SiSCl2)4. Despite the double bond rule, stable organosilanethiones RR'Si=S have been made thanks to the stabilising mechanism of intermolecular coordination via an amine group.[31]
Silicon nitride, Si3N4, may be formed by directly reacting silicon with nitrogen above 1300 °C, but a more economical means of production is by heating silica and coke in a stream of nitrogen and hydrogen gas at 1500 °C. It would make a promising ceramic if not for the difficulty of working with and sintering it: chemically, it is near-totally inert, and even above 1000 °C it keeps its strength, shape, and continues to be resistant to wear and corrosion. It is very hard (9 on the Mohs hardness scale), dissociates only at 1900 °C at 1 atm, and is quite dense (density 3.185 g/cm3), because of its compact structure similar to that of phenacite (Be
2SiO
4). A similar refractory material is Si2N2O, formed by heating silicon and silica at 1450 °C in an argon stream containing 5% nitrogen gas, involving 4-coordinate silicon and 3-coordinate nitrogen alternating in puckered hexagonal tilings interlinked by non-linear Si–O–Si linkages to each other.[31]
Reacting silyl halides with ammonia or alkylammonia derivatives in the gaseous phase or in ethanolic solution produces various volatile silylamides, which are silicon analogues of the amines:[31]
- 3 SiH3Cl + 4 NH3 → N(SiH3)3 + 3 NH4Cl
- SiH3Br + 2 Me2NH → SiH3NMe2 + Me2NH2Br
- 4 SiH3I + 5 N2H4 → (SiH3)2NN(SiH3)2 + 4 N2H5I
Many such compounds have been prepared, the only known restriction being that the nitrogen is always tertiary, and species containing the SiH–NH group are unstable at room temperature. The stoichiometry around the nitrogen atom in compounds such as N(SiH3)3is planar, which has been attributed to a pπ–dπ interaction between a lone pair on nitrogen and an empty dπ orbital on silicon. Similarly, trisilylamines are weaker as ligands than their carbon analogues, the tertiary amines, although substitution of some SiH3 groups by CH3 groups mitigates this weakness. For example, N(SiH3)3 does not form an adduct with BH3 at all, while MeN(SiH3)2 and Me2NSiH3 form adducts at low temperatures that decompose upon warming. Some silicon analogues of imines, with a Si=N double bond, are known: the first found was But2Si=N–SiBut3, which was discovered in 1986.[31]

Silicon carbide (SiC) was first made by Edward Goodrich Acheson in 1891, who named it carborundum to reference its intermediate hardness and abrasive power between diamond (an allotrope of carbon) and corundum (aluminium oxide). He soon founded a company to manufacture it, and today about one million tonnes are produced each year.[32] Silicon carbide exists in about 250 crystalline forms.[33] The polymorphism of SiC is characterized by a large family of similar crystalline structures called polytypes. They are variations of the same chemical compound that are identical in two dimensions and differ in the third. Thus they can be viewed as layers stacked in a certain sequence.[34] It is made industrially by reduction of quartz sand with excess coke or anthracite at 2000–2500 °C in an electric furnace:[32]
- SiO2 + 2 C → Si + 2 CO
- Si + C → SiC
It is the most thermally stable binary silicon compound, only decomposing through loss of silicon starting from around 2700 °C. It is resistant to most aqueous acids, phosphoric acid being an exception. It forms a protective layer of silicon dioxide on the surface and hence only oxidises appreciably in air above 1000 °C; removal of this layer by molten hydroxides or carbonates leads to quick oxidation. Silicon carbide is rapidly attacked by chlorine gas, which forms SiCl4 and carbon at 100 °C and SiCl4 and CCl4 at 1000 °C. It is mostly used as an abrasive and a refractory material, as it is chemically stable and very strong, and it fractures to form a very sharp cutting edge. It is also useful as an intrinsic semiconductor, as well as an extrinsic semiconductor upon being doped.[32] In its diamond-like behavior it serves as an illustration of the chemical similarity between carbon and silicon.[35]
Organosilicon compounds
[edit]
Because the Si–C bond is close in strength to the C–C bond, organosilicon compounds tend to be markedly thermally and chemically stable. For example, tetraphenylsilane (SiPh4) may be distilled in air even at its boiling point of 428 °C, and so may its substituted derivatives Ph3SiCl and Ph2SiCl2, which boil at 378 °C and 305 °C respectively. Furthermore, since carbon and silicon are chemical congeners, organosilicon chemistry shows some significant similarities with carbon chemistry, for example in the propensity of such compounds for catenation and forming multiple bonds.[35] However, significant differences also arise: since silicon is more electropositive than carbon, bonds to more electronegative elements are generally stronger with silicon than with carbon, and vice versa. Thus the Si–F bond is significantly stronger than even the C–F bond and is one of the strongest single bonds, while the Si–H bond is much weaker than the C–H bond and is readily broken. Furthermore, the ability of silicon to expand its octet is not shared by carbon, and hence some organosilicon reactions have no organic analogues. For example, nucleophilic attack on silicon does not proceed by the SN2 or SN1 processes, but instead goes through a negatively charged true pentacoordinate intermediate and appears like a substitution at a hindered tertiary atom. This works for silicon, unlike for carbon, because the long Si–C bonds reduce the steric hindrance and the d-orbital of silicon is geometrically unconstrained for nucleophilic attack, unlike for example a C–O σ* antibonding orbital. Nevertheless, despite these differences, the mechanism is still often called "SN2 at silicon" for simplicity.[36]
One of the most useful silicon-containing groups is trimethylsilyl, Me3Si–. The Si–C bond connecting it to the rest of the molecule is reasonably strong, allowing it to remain while the rest of the molecule undergoes reactions, but is not so strong that it cannot be removed specifically when needed, for example by the fluoride ion, which is a very weak nucleophile for carbon compounds but a very strong one for organosilicon compounds. It may be compared to acidic protons; while trisilylmethyl is removed by hard nucleophiles instead of bases, both removals usually promote elimination. As a general rule, while saturated carbon is best attacked by nucleophiles that are neutral compounds, those based on nonmetals far down on the periodic table (e.g. sulfur, selenium, or iodine), or even both, silicon is best attacked by charged nucleophiles, particularly those involving such highly electronegative nonmetals as oxygen, fluorine, or chlorine. For example, enolates react at the carbon in haloalkanes, but at the oxygen in silyl chlorides; and when trimethylsilyl is removed from an organic molecule using hydroxide as a nucleophile, the product of the reaction is not the silanol as one would expect from using carbon chemistry as an analogy, because the siloxide is strongly nucleophilic and attacks the original molecule to yield the silyl ether hexamethyldisiloxane, (Me3Si)2O. Conversely, while the SN2 reaction is mostly unaffected by the presence of a partial positive charge (δ+) at the carbon, the analogous "SN2" reaction at silicon is so affected. Thus, for example, the silyl triflates are so electrophilic that they react 108 to 109 times faster than silyl chlorides with oxygen-containing nucleophiles. Trimethylsilyl triflate is in particular a very good Lewis acid and is used to convert carbonyl compounds to acetals and silyl enol ethers, reacting them together analogously to the aldol reaction.[36]
Si–C bonds are commonly formed in three ways. In the laboratory, preparation is often carried out in small quantities by reacting tetrachlorosilane (silicon tetrachloride) with organolithium, Grignard, or organoaluminium reagents, or by catalytic addition of Si–H across C=C double bonds. The second route has the drawback of not being applicable to the most important silanes, the methyl and phenyl silanes. Organosilanes are made industrially by directly reacting alkyl or aryl halides with silicon with 10% by weight metallic copper as a catalyst. Standard organic reactions suffice to produce many derivatives; the resulting organosilanes are often significantly more reactive than their carbon congeners, readily undergoing hydrolysis, ammonolysis, alcoholysis, and condensation to form cyclic oligomers or linear polymers.[35]
Differences with carbon
[edit]Silicon shows clear differences from carbon. For example, organic chemistry has very few analogies with silicon chemistry, while silicate minerals have a structural complexity unseen in oxocarbons.[37] Silicon tends to resemble germanium far more than it does carbon, and this resemblance is enhanced by the d-block contraction, resulting in the size of the germanium atom being much closer to that of the silicon atom than periodic trends would predict.[30] Nevertheless, there are still some differences because of the growing importance of the divalent state in germanium compared to silicon, which result in germanium being significantly more metallic than silicon. Additionally, the lower Ge–O bond strength compared to the Si–O bond strength results in the absence of "germanone" polymers that would be analogous to silicone polymers.[10]
Silicone polymers
[edit]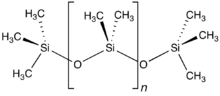
The word "silicone" was first used by Frederic Kipping in 1901. He invented the word to illustrate the similarity of chemical formulae between Ph2SiO and benzophenone, Ph2CO, although he also stressed the lack of chemical resemblance due to the polymeric structure of Ph2SiO, which is not shared by Ph2CO.[35]
Silicones may be considered analogous to mineral silicates, in which the methyl groups of the silicones correspond to the isoelectronic O− of the silicates.[35] They are quite stable to extreme temperatures, oxidation, and water, and have useful dielectric, antistick, and antifoam properties. Furthermore, they are resistant over long periods of time to ultraviolet radiation and weathering, and are inert physiologically. They are fairly unreactive, but do react with concentrated solutions bearing the hydroxide ion and fluorinating agents, and occasionally, may even be used as mild reagents for selective syntheses. For example, (Me3Si)2O is valuable for the preparation of derivatives of molybdenum and tungsten oxyhalides, converting a tungsten hexachloride suspension in dichloroethane solution quantitatively to WOCl4 in under an hour at room temperature, and then to yellow WO2Cl2 at 100 °C in light petroleum at a yield of 95% overnight.[35]
See Also
[edit]References
[edit]- ^ a b c d e f g Greenwood & Earnshaw 1997, p. 330.
- ^ "New Type of Zero-Valent Tin Compound". Chemistry Europe. 27 August 2016.
- ^ Ram, R. S.; et al. (1998). "Fourier Transform Emission Spectroscopy of the A2D–X2P Transition of SiH and SiD" (PDF). J. Mol. Spectr. 190 (2): 341–352. doi:10.1006/jmsp.1998.7582. PMID 9668026.
- ^ Stapf, André; Gondek, Christoph; Kroke, Edwin; Roewer, Gerhard (2019), Yang, Deren (ed.), "Wafer Cleaning, Etching, and Texturization", Handbook of Photovoltaic Silicon, Berlin, Heidelberg: Springer Berlin Heidelberg, pp. 311–358, doi:10.1007/978-3-662-56472-1_17, ISBN 978-3-662-56471-4, S2CID 226945433, retrieved 2021-03-07
- ^ a b Grabmaier, J. (1982). Silicon Chemical Etching. Berlin, Heidelberg: Springer Berlin Heidelberg. ISBN 978-3-642-68765-5. OCLC 840294227.
- ^ a b c d e f g Greenwood & Earnshaw 1997, p. 331.
- ^ Greenwood and Earnshaw, p. 331–335
- ^ Kaupp, Martin (1 December 2006). "The role of radial nodes of atomic orbitals for chemical bonding and the periodic table" (PDF). Journal of Computational Chemistry. 28 (1): 320–325. doi:10.1002/jcc.20522. PMID 17143872. S2CID 12677737. Retrieved 14 October 2016.
- ^ a b c King, pp. 43–44
- ^ a b Greenwood & Earnshaw 1997, pp. 374.
- ^ Kaupp, Martin (2007). "The role of radial nodes of atomic orbitals for chemical bonding and the periodic table". Journal of Computational Chemistry. 28 (1): 320–325. doi:10.1002/jcc.20522. ISSN 0192-8651. PMID 17143872.
- ^ Greenwood & Earnshaw 1997, p. 328.
- ^ Greenwood & Earnshaw 1997, p. 335-337.
- ^ King, pp. 45–47
- ^ E. Wiber, Alfred Stock and the Renaissance of Inorganic Chemistry," Pure Appl. Chem., Vol. 49 (1977) pp. 691–700.
- ^ J. W. Mellor, "A Comprehensive Treatise on Inorganic and Theoretical Chemistry," Vol. VI, Longman, Green and Co. (1947) pp. 223–227.
- ^ W. W. Porterfield, Inorganic Chemistry: A Unified Approach, 2nd Ed.", Academic Press (1993), p. 219.
- ^ Inorganic Chemistry, Holleman-Wiberg, John Wiley & Sons (2001) p. 844.
- ^ King, p. 47
- ^ Miller, R.D.; Michl, J. (1989). "Polysilane high polymers". Chemical Reviews. 89 (6): 1359. doi:10.1021/cr00096a006.
- ^ a b c d Greenwood & Earnshaw 1997, pp. 340.
- ^ a b c King, p. 48
- ^ Greenwood & Earnshaw 1997, p. 342-347.
- ^ a b c Greenwood & Earnshaw 1997, p. 342.
- ^ a b c d e Greenwood & Earnshaw 1997, p. 347.
- ^ Geological Survey (U.S.) (1975). Geological Survey professional paper.
- ^ Korzhinsky, M.A.; Tkachenko, S.I.; Shmulovich, K.I.; Steinberg, G.S. (1995). "Native AI and Si formation" (PDF). Nature. 375 (6532): 544. Bibcode:1995Natur.375..544K. doi:10.1038/375544a0. ISSN 0028-0836. S2CID 39954119.
- ^ Cordua, Courtesy of Dr Bill (1998-01-10), English: PDF file entitled: "Silicon, Silica, Silicates and Silicone" (PDF), archived from the original (PDF) on 2016-04-18, retrieved 2016-03-29
- ^ Greenwood & Earnshaw 1997, p. 347-359.
- ^ a b Greenwood & Earnshaw 1997, pp. 359–361.
- ^ a b c d Greenwood & Earnshaw 1997, p. 359.
- ^ a b c Greenwood & Earnshaw 1997, p. 334.
- ^ Cheung, Rebecca (2006). Silicon carbide microelectromechanical systems for harsh environments. Imperial College Press. p. 3. ISBN 978-1-86094-624-0.
- ^ Morkoç, H.; Strite, S.; Gao, G.B.; Lin, M.E.; Sverdlov, B.; Burns, M. (1994). "Large-band-gap SiC, III–V nitride, and II–VI ZnSe-based semiconductor device technologies". Journal of Applied Physics. 76 (3): 1363. Bibcode:1994JAP....76.1363M. doi:10.1063/1.358463.
- ^ a b c d e f Greenwood & Earnshaw 1997, p. 361.
- ^ a b Clayden, pp. 668–77
- ^ Greenwood & Earnshaw 1997, pp. 327–328.
Category:Silicon Category:Silicon compounds Category:Chemical compounds by element





