User:Bethany Thames/Sandbox
Auditory fatigue is defined as a temporary loss of hearing after exposure to sound. This results in a temporary shift of the auditory threshold known as a temporary threshold shift, or TTS. The damage can become permanent (permanent threshold shift, PTS) if sufficient recovery time is not allowed for before continued sound exposure. When the hearing loss is rooted from a traumatic occurrence, it may be classified as noise-induced hearing loss, or NIHL.
There are two main types of auditory fatigue, short-term and long-term.[1] These are distinguished from each other by several characteristics listed individually below.
Short-term fatigue
- full recovery from TTS can be achieved in approximately two minutes
- the TTS is relatively independent of exposure duration[1][2]
- TTS is maximal at the exposure frequency
Long-term fatigue
- recovery requires a minimum of several minutes but can take up to days
- dependent on exposure duration and noise level[1][2]
Physiology
[edit]Affected anatomy
[edit]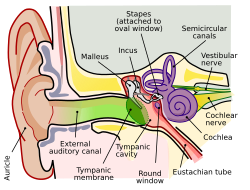
Note: The complete anatomy of the ear is extensive, and can be divided into the inner ear and outer ear.
The remainder of this article mainly references the cochlea, outer hair cells, and Organ of Corti.
In general, structural damages to any anatomical parts of the ear can cause hearing-related problems. Usually, minor bending of the stereocilia (inner ear) is associated with temporary hearing loss and is involved in auditory fatigue. Complete loss of the stereocilia causes permanent hearing damage and is more associated with noise-induced hearing loss and other auditory diseases.
The outer hair cells outer hair cells, or OHCs, can be thought of as microamplifiers that provide stimulation to the inner hair cells. The OHCs are the most fragile of the hair cells, hence their involvement in auditory fatigue and other hearing impairments.

|

|

|
| Depiction of inner ear showing Cochlea | Depiction of Cochlea showing Organ of Corti | Organ of Corti showing hair cells |
Affected mechanisms
[edit]Traveling wave theory
[edit]Temporary threshold shifts related to auditory fatigue are related to the amplitude of a stimulus-driven traveling wave.[3] This is believed to be true because the vibration propagated by the active process is not usually at the center of the maximum amplitude of this wave. Instead, it is located much further down and the differences associated between them explain the shift in threshold.[1] This TTS that is experienced is the exhaustion of the active system described below located at the locus of the traveling wave driven by the cochlear amplifier.[3] Auditory fatigue can be explained by the relative activity of the active process at low-level stimulation (<30dB).[1]
Classical passive system
[edit]There are two different systems associated with the mechanics of the cochlea: the classical passive system and an active process. The passive system works to stimulate the inner hair cells directly and works at levels above 40dB.[3] At stimulation levels that prevent the excitation of the passive system, prolonged noise exposure results in a decrease in the loudness heard over time, even when the actual intensity of the noise has not changed.[1] This is caused by the exhaustion of the active system.
Active process
[edit]The active system is also known as the cochlear amplifier. This amplification increases vibrations of the basilar membrane through energy obtained from the Organ of Corti.[3] As the stimulation increases, it is assumed that the basilar membrane displacement caused by the traveling wave becomes continually more basal in regards to the cochlea.[4] A sustained low-level stimulus can cause an energetic exhaustion of the active system which in turn prevents the passive system from activating.
Excessive vibrations
[edit]Currently it is believed that auditory fatigue and NIHL are related to excessive vibrations of the inner ear which may cause structural damages.[5][6][7] Metabolic activity is required in order to maintain the electrochemical gradients used in mechano-electrical and electro-mechanical transduction during noise exposure and sound recognition.[5] The metabolic activity is associated with active displacements which are components of the sound-induced vibration involving prestin, a motor protein that causes OHC motility.[5] With excess vibrations, the metabolic energy required is increased.
In addition, these extra vibrations can cause the formation of free radicals known as reactive oxygen species or ROS. [8][9] Elevated levels of ROS continue to increase the metabolic demands of the system. These increasing demands fatigue the system and eventually lead to structural damages to the Organ of Corti.[5][10]
Recovery
[edit]In all cases of auditory fatigue, sufficient recovery time should allow for full correction of the hearing impairment.[1] This will return threshold levels to their baseline values. However, there is currently no way to estimate the amount of time needed to recover from auditory fatigue because it is not usually detectable until after the injury has already occurred. However, studies that measured recovery time have noted that the amount of time required is related to the magnitude of the initial hearing loss.[11] The most significant recovery was observed to occur during the first 15 minutes following the cessation of the noise exposure.[12][13] It is when sufficient recovery time is not allotted that effects become permanent causing acquired noise-induced hearing loss.[11] Up to 120 minutes of recovery time can be required of noises of only 95 dB.[11] For comparison, common items that can produce noise at this level are motorcycles and subways.[14]
Protective measures
[edit]Toughening and energy spread
[edit]Two protective measures have been investigated related to the amount of noise exposure and the duration of that exposure. Although these would be hard to regulate in spontaneous occurrences, they may could have a positive effect on work conditions if guidelines could be set for machining times or for other systems that produce loud noises over a long period of time. The toughening effect is put in place by increasing the system's resistance to noise over time.[15] Currently, the specific mechanisms that cause the cochlear toughening are not known. However, the OHCs and related processes are known to play a role.[16]The other idea is to spread a given amount of energy to the system over a longer amount of time. This would allow recovery processes to take place during the quiet interludes that are gained by increasing the exposure duration.[15] So far, studies have not shown a direct correlation between the amount of toughening and the amount of threshold shift experienced.[15] This suggests that even a toughened cochlea may not be completely protected.
Substances
[edit]Both furosemide and salicylic acid are considered ototoxic at certain doses. However, research has been done to determine their ability to protect against auditory fatigue and permanent damage through toughening phenomena, a state described by reduced active cochlear displacements. Limited research has been done with these two substances in terms of protective drug regimes because of their associated risks. However, both have shown positive results in reducing auditory fatigue by the decrease in ROS formation through individual mechanisms described below.[5][17]
Furosemide injections prior to noise exposure have been shown to decrease the endocochlear potential.[18] This decrease results in a reduction of active cochlear displacements and it is believed that the protection by furosemide stems from the limitation of excessive vibrations while the cochlear amplifier is depressed.[19]
Salicylic acid competitively interferes with anion binding to OHC prestin which thereby reduces motility. This reduction in active displacement is again associated with depression of the cochlear amplifier which decreases the excessive vibrations experienced during noise-exposure.[6][7][8][10]
Vitamins A, C and E have been shown to be 'free radical scavengers' by studies looking for protective tendencies of antioxidants.[20] In addition, NAC, or N-acetyl-L-cysteine (acetylcysteine), has been shown to reduce reactive oxygen species formation associated with the excessive vibrations induced by the noise exposure.[21][22][9]
Limitations
[edit]Although auditory fatigue and NIHL protective measures would be helpful for those who are constantly exposed to long and loud noises, current research is limited due to the negative associations with the substances.[5] Furosemide is used in congestive heart failure treatments because of its diuretic properties. Salicylic acid is a compound most frequently used in anti-acne washes, but is also an anticoagulant. Further uses of these substances would need to be personalized to the individual and only under close monitoring. Antioxidants do not have these negative effects and therefore are the most commonly researched substance for the purpose of protecting against auditory fatigue.[5] However, at this time there has been no marketed application. In addition, no synergistic relationship between the drugs on the degree of reduction of auditory fatigue have been discovered at this time.[23]
Risk increasing factors
[edit]- Physical exercise
- Heat exposure
- Workload
- Smoking
- Ototoxic chemicals
There are several factors that may not be harmful to the auditory system by themselves, but when paired with an extended noise exposure duration have been shown to increase the risk of auditory fatigue. This is important because humans will remove themselves from a noisy environment if it passes their pain threshold.[11] However, when paired with other factors that may not physically recognizable as damaging, TTS may be greater even with less noise exposure. One such factor is physical exercise. Although this is generally good for the body, combined noise exposure during highly physical activities was shown to produce a greater TTS than just the noise exposure alone.[24][25] This could be related to the amount of reactive oxygen species being produced by the excessive vibrations further increasing the metabolic activity required, which is already increased during physical exercise. However, a person can decrease their susceptibility to TTS by improving their cardiovascular fitness overall.[11]
Heat exposure is another risk factor. As blood temperature rises, TTS increases when paired with high-frequency noise exposure.[11] It is hypothesized that hair cells for high-frequency transduction require a greater oxygen supply than others, and the two simultaneous metabolic can deplete any oxygen reserves of the cochlea.[26] In this case, the auditory system undergoes temporary changes caused by a decrease in the oxygen tension of the cochlear endolymph that leads to vasoconstriction of the local vessels.[27] Further research could be done to see if this is a reason for the increased TTS during physical exercise that is during continued noise-exposure as well.
Another factor that may not show signs of being harmful is the current workload of a person. Exposure to noise greater than 95dB in individuals with heavy workloads was shown to cause severe TTS.[11] In addition, the workload was a driving factor in the amount of recovery time required to return threshold levels to their baselines.[11] In contrast, a factor that is harmful to the body, but not normally directly to the auditory system is smoking. This common lifestyle habit has also been shown to increase temporary threshold shifts that occur from noise exposure damages.
There are some factors that are known to directly effect the auditory system. Contact with ototoxic chemicals such as styrene, toluene and carbon disulfide heighten the risk of auditory damages.[11] Those individuals in work environments are more likely to experience the noise and chemical combination that can increase the likelihood of auditory fatigue.[9][28] Individually, styrene is known to cause structural damages of the cochlea without actually interfering with functional capabilities.[9] This explains the synergistic interaction between noise and styrene because the cochlea will be increasingly damaged with the excessive vibrations of the noise plus the damage caused by the chemical itself. Specifically, noise damage typically damages the first layer of the outer hair cells. The combined effects of styrene and noise exposure shows damages to all three rows instead, reinforcing previous results.[9] Also, the combined effects of these chemicals and the noise produce greater auditory fatigue than when an individual is exposed to one factor immediately followed by the next.[9]
It is important to understand that noise exposure itself is the main influential factor in threshold shifts and auditory fatigue, but that individuals may be at greater risk when synergistic effects take place during interactions with the above factors.[11]
Experimental Studies
[edit]Human
[edit]Dolphin
[edit]- Finneran[31]
Rodents
[edit]Mice
[edit]- Groschel[32]
Rats
[edit]- Chen[9]
Guinea-pigs
[edit]Chinchillas
[edit]- Hamernik[15]
References
[edit]- ^ a b c d e f g Charron, S., & Botte, M. C. (1988). Frequency-selectivity in loudness adaptation and auditory fatigue. [Article]. Journal of the Acoustical Society of America, 83(1), 178-187.
- ^ a b Hirsh IJ, Bilger RC, Burns W. Auditory-Threshold Recovery after Exposures to Pure Tones. The Journal of the Acoustical Society of America. 1955;27(5):1013-1013.
- ^ a b c d Davis H. An active process in cochlear mechanics. Hearing Research. 1983;9(1):79-90.
- ^ McFadden D, Plattsmier H. Exposure-induced loudness shifts and threshold shifts. New Perspectives in Noise-induced Hearing Loss. 1982:363-374.
- ^ a b c d e f g Adelman, C., Perez, R., Nazarian, Y., Freeman, S., Weinberger, J., & Sohmer, H. (2010). Furosemide Administered Before Noise Exposure Can Protect the Ear. [Article]. Annals of Otology Rhinology and Laryngology, 119(5), 342-349.
- ^ a b Ou HC, Bohne BA, Harding GW. Noise damage in the C57BL/CBA mouse cochlea. Hearing Research. 2000;145(1-2):111-122.
- ^ a b Wang Y, Hirose K, Liberman MC. Dynamics of Noise-Induced Cellular Injury and Repair in the Mouse Cochlea. JARO - Journal of the Association for Research in Otolaryngology. 2002;3(3):248-268.
- ^ a b Ohlemiller KK, Wright JS, Dugan LL. Early Elevation of Cochlear Reactive Oxygen Species following Noise Exposure. Audiology and Neurotology. 1999;4(5):229-236.
- ^ a b c d e f g Chen G-D, Henderson D. Cochlear injuries induced by the combined exposure to noise and styrene. Hearing Research. 2009;254(1-2):25-33. Cite error: The named reference "Chen 2009" was defined multiple times with different content (see the help page).
- ^ a b Henderson D, Bielefeld E, Harris K, Hu B. The role of oxidative stress in noise-induced hearing loss. Ear Hear. 2006;27:1 - 19.
- ^ a b c d e f g h i j Chen C-J, Dai Y-T, Sun Y-M, Lin Y-C, Juang Y-J. Evaluation of Auditory Fatigue in Combined Noise, Heat and Workload Exposure. Industrial Health. 2007;45(4):527-534.
- ^ Ward WD. Temporary threshold shift and damage-risk criteria for intermittent noise exposures. Journal of the Acoustical Society of America. 1970(48):561-574.
- ^ Ward WD. Recovery from high values of temporary threshold shift. Journal of the Acoustical Society of America. 1970(32):497-500.
- ^ <http://condor.admin.ccny.cuny.edu/~ms6785/page4.html>
- ^ a b c d <Hamernik RP, Ahroon WA. Interrupted noise exposures: Threshold shift dynamics and permanent effects. The Journal of the Acoustical Society of America. 1998;103(6):3478-3488. Cite error: The named reference "Hamernik 98" was defined multiple times with different content (see the help page).
- ^ Zheng X-Y, Henderson D, McFadden SL, Hu B-H. The role of the cochlear efferent system in acquired resistance to noise-induced hearing loss. Hearing Research. 1997;104(1-2):191-203.
- ^ Adelman C, Freeman S, Paz Z, Sohmer H. Salicylic acid injection before noise exposure reduces permanent threshold shift. Audiol Neurootol. 2008;13:266 - 272.
- ^ Ruggero M, Rich N. Furosemide alters organ of corti mechanics: evidence for feedback of outer hair cells upon the basilar membrane. J Neurosci. 1991;11:1057 - 1067.
- ^ Ikeda K, Morizono T. Effect of albumin-bound furosemide on the endocochlear potential of the chinchilla. Alleviation of furosemide-induced ototoxicity. Arch Otolaryngol Head Neck Surg. 1989;115:500 - 502.
- ^ Le Prell CG, Hughes LF, Miller JM. Free radical scavengers vitamins A, C, and E plus magnesium reduce noise trauma. Free Radical Biology and Medicine. 2007;42(9):1454-1463.
- ^ Bielefeld E, Kopke R, Jackson R, Coleman J, Liu J, Henderson D. Noise protection with N-acetyl-l-cysteine (NAC) using a variety of noise exposures, NAC doses, and routes of administration. Acta Otolaryngol. 2007;127:914 - 919.
- ^ Kopke RD, Jackson RL, Coleman JKM, Liu J, Bielefeld EC, Balough BJ. NAC for noise: From the bench top to the clinic. Hearing Research. 2007;226(1-2):114-125.
- ^ Tamir S, Adelman C, Weinberger J, Sohmer H. Uniform comparison of several drugs which provide protection from noise induced hearing loss. Journal of Occupational Medicine and Toxicology. 2010;5(1):26.
- ^ Lindgren F, Axelsson A. The Influence of Physical Exercise on Susceptibility to Noise-Induced Temporary Threshold Shift. Scandinavian Audiology. 1988;17(1):11-17
- ^ <Miani C, Bertino G, Francescato M, di Prampero P, Staffieri A. Temporary Threshold Shift Induced by Physical Exercise. Scandinavian Audiology. 1996;25(3):179-186.
- ^ Miller J, Ren T, Dengerink H, Nuttall A. Cochlear blood flow changes with short sound stimulation. Scientific Basis of Noise-Induced Hearing Loss. 1996:95-109.
- ^ Axelsson A, Vertes D, Miller J. Immediate Noise Effects on Cochlear Vasculature in the Guinea Pig. Acta Oto-Laryngol. 1981;91(1-6):237-246.
- ^ Mizoue T, Miyamoto T, Simizu T. Combined effect of smoking and occupational exposure to noise on hearing loss in steel factory workers. Occupational and Environmental Medicine. 2003; 60:56-59.
- ^ Lin, C. Y., Wu, J. L., Shih, T. S., Tsai, P. J., Sun, Y. M., & Guo, Y. L. (2009). Glutathione S-transferase M1, T1, and P1 polymorphisms as susceptibility factors for noise-induced temporary threshold shift. [Article]. Hearing Research, 257(1-2), 8-15. doi: 10.1016/j.heares.2009.07.008
- ^ Melnick, W. (1991). HUMAN TEMPORARY THRESHOLD SHIFT (TTS) AND DAMAGE RISK. [Article]. Journal of the Acoustical Society of America, 90(1), 147-154.
- ^ Finneran, J. J., & Schlundt, C. E. (2010). Frequency-dependent and longitudinal changes in noise-induced hearing loss in a bottlenose dolphin (Tursiops truncatus) (L). Journal of the Acoustical Society of America, 128(2), 567-570. doi: 10.1121/1.3458814
- ^ Groschel, M., Gotze, R., Ernst, A., & Basta, D. (2010). Differential Impact of Temporary and Permanent Noise-Induced Hearing Loss on Neuronal Cell Density in the Mouse Central Auditory Pathway. [Article]. Journal of Neurotrauma, 27(8), 1499-1507. doi: 10.1089/neu.2009.1246
- ^ Fetoni, A. R., Mancuso, C., Eramo, S. L. M., Ralli, M., Piacentini, R., Barone, E., et al. (2010). IN VIVO PROTECTIVE EFFECT OF FERULIC ACID AGAINST NOISE-INDUCED HEARING LOSS IN THE GUINEA-PIG. [Article]. Neuroscience, 169(4), 1575-1588. doi: 10.1016/j.neuroscience.2010.06.022
- ^ Gourevitch, B., Doisy, T., Avillac, M., & Edeline, J. M. (2009). Follow-up of latency and threshold shifts of auditory brainstem responses after single and interrupted acoustic trauma in guinea pig. [Article]. Brain Research, 1304, 66-79. doi: 10.1016/j.brainres.2009.09.041
- ^ Chen, Y. S., Tseng, F. Y., Lin, K. N., Yang, T. H., Lin-Shiau, S. Y., & Hsu, C. J. (2008). Chronologic Changes of Nitric Oxide Concentration in the Cochlear Lateral Wall and Its Role in Noise-Induced Permanent Threshold Shift. [Article]. Laryngoscope, 118(5), 832-836. doi: 10.1097/MLG.0b013e3181651c24
- ^ Yamashita, D., Minami, S. B., Kanzaki, S., Ogawa, K., & Miller, J. M. (2008). Bcl-2 genes regulate noise-induced hearing loss. [Article]. Journal of Neuroscience Research, 86(4), 920-928. doi: 10.1002/jnr.21533
