Codeine methylbromide
 | |
| Clinical data | |
|---|---|
| Other names | Codeine bromomethylate, 125-27-9, DEA No. 9070 |
| AHFS/Drugs.com | Monograph |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
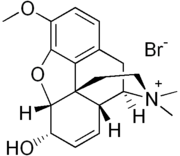
| Formula | C19H24BrNO3 |
| Molar mass | 394.309 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Codeine methylbromide (Eucodin) is the bromomethane (methylbromide) salt of codeine. Its possession is prohibited in many jurisdictions. It is considered a Schedule I controlled substance in the United States, with a DEA ACSCN of 9070 and nil annual aggregate manufacturing quota.[1] as of 2014. As it is used in a different way than basic salts of codeine like the phosphate or hydrochloride owing to its below-mentioned dual action, it is considered to be a different drug related to codeine rather than merely a salt of it in many contexts.[2]
Also known by the genericised trade name eucodeine, and the salt name also sometimes given as methobromide, this drug was first synthesised in Austria-Hungary in 1903. As it is a bromide in addition to a codeine salt, it has a dual mechanism of action and is indicated for pain with insomnia or nervousness and violent coughing.[2] This codeine-based bromide also has morphine, dihydrocodeine, dihydromorphine, hydromorphone, isocodeine, hydrocodone, and other such analogues; also, there are codeine-based barbiturates and salicylates.[3]
References
[edit]This article needs additional citations for verification. (March 2017) |
- ^ "Conversion Factors for Controlled Substances". Diversion Control Division, Drug Enforcement Administration. U.S. Department of Justice.
- ^ a b Lowry WT, Garriott JC (1979). Forensic Toxicology: Controlled Substances and Dangerous Drugs. Boston, MA: Springer US. pp. 176–177. ISBN 978-1-4684-3444-6.
- ^ The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (13th ed.). Whitehouse Station, N.J.: Merck. 2001. ISBN 978-0-911910-13-1.
