β-Pinene
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
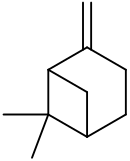
6,6-Dimethyl-2-methylidenebicyclo[3.1.1]heptane
Pin-2(10)-ene | |||
| Other names
6,6-Dimethyl-2-methylenebicyclo[3.1.1]heptane
2(10)-Pinene Nopinene Pseudopinene | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.004.430 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C10H16 | |||
| Molar mass | 136.238 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Density | 0.872 g/mL | ||
| Melting point | −61.54 °C; −78.77 °F; 211.61 K[1] | ||
| Boiling point | 165–167 °C; 329–332 °F; 438–440 K[2] | ||
| Thermochemistry | |||
Std enthalpy of
combustion (ΔcH⦵298) |
−6214.1±2.9 kJ/mol[1] | ||
| Hazards | |||
| GHS labelling: | |||
   
| |||
| Danger | |||
| H226, H304, H315, H317, H410 | |||
| P210, P233, P240, P241, P242, P243, P261, P264, P272, P273, P280, P301+P310, P302+P352, P303+P361+P353, P321, P331, P332+P313, P333+P313, P362, P363, P370+P378, P391, P403+P235, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 36 °C (97 °F; 309 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
β-Pinene is a monoterpene, an organic compound found in plants. It is the less abundant of the two isomers of pinene, the other being α-pinene.[3] It is a colorless liquid soluble in alcohol, but not water. It has a woody-green pine-like smell.
β-Pinene is one of the most abundant compounds released by forest trees.[4] If oxidized in air, the allylic products of the pinocarveol and myrtenol family prevail.[5]
Sources
[edit]Many plants from many botanical families contain the compound, including:
- Cuminum cyminum[6][7]
- Humulus lupulus[8]
- Pinus pinaster[5]
- Clausena anisata
- Cannabis sativa[9]
- Piper nigrum[10]
- Myristica fragans .[10]
- Citrus aurantiifolia[10]
- Pistacia lentiscus[10]
The clear compound is produced by distillation of turpentine oils.[11]
Uses
[edit]β-Pinene is usedin the production of other aroma compounds. It converts to myrcene upon heating at 500 °C. Nerol is obtained by careful fractional distillation of crude nerol from myrcene[12]).[13]
Reaction with formaldehyde (Prins reaction) converts β-pinene to nopol. When nopol is acetylated, the result is nopyl acetate, which is used as fragrance material.[11][14]
References
[edit]- ^ a b "β-Pinene". National Institute of Standards and Technology. Retrieved January 29, 2018.
- ^ "(−)-β-Pinene". Sigma-Aldrich. Retrieved January 29, 2018.
- ^ Charles S. Sell (2013), "Terpenoids", in Arza Seidel; et al. (eds.), Kirk-Othmer Chemical Technology of Cosmetics, John Wiley & Sons, pp. 247–374, ISBN 978-1-118-40692-2
- ^ Geron, C., et al. (2000). A review and synthesis of monoterpene speciation from forests in the United States. Atmospheric Environment 34(11), 1761-81.
- ^ a b Neuenschwander, U.; Meier, E.; Hermans, I. (2011). "Peculiarities of β-pinene autoxidation". ChemSusChem. 4 (11): 1613–21. doi:10.1002/cssc.201100266. PMID 21901836.
- ^ Li, Rong; Jiang, Zi-Tao (2004). "Chemical composition of the essential oil of Cuminum cyminum L. From China". Flavour and Fragrance Journal. 19 (4): 311–313. doi:10.1002/ffj.1302.
- ^ Wang, L.; Wang, Z.; Zhang, H.; Li, X.; Zhang, H. (2009). "Ultrasonic nebulization extraction coupled with headspace single drop microextraction and gas chromatography-mass spectrometry for analysis of the essential oil in Cuminum cyminum L". Analytica Chimica Acta. 647 (1): 72–7. doi:10.1016/j.aca.2009.05.030. PMID 19576388.
- ^ Tinseth, G. The Essential Oil of Hops: Hop Aroma and Flavor in Hops and Beer. Archived 2013-11-11 at the Wayback Machine Brewing Techniques January/February 1994. Accessed July 21, 2010.
- ^ Hillig, Karl W (October 2004). "A chemotaxonomic analysis of terpenoid variation in Cannabis". Biochemical Systematics and Ecology. 32 (10): 875–891. doi:10.1016/j.bse.2004.04.004. ISSN 0305-1978.
- ^ a b c d Santana de Oliveira, Mozaniel (2022). Essential oils: applications and trends in food science and technology. Cham, Switzerland: Springer. ISBN 978-3-030-99476-1.
- ^ a b Surburg, Horst; Panten, Johannes (2016). Common fragrance and flavor materials: preparation, properties and uses (6. completely revised and updated ed.). Weinheim: Wiley-VCH Verlag GmbH & Co. KGaA. ISBN 978-3-527-33160-4.
- ^ Opdyke, D. L. J. (2013-10-22). Monographs on Fragrance Raw Materials: A Collection of Monographs Originally Appearing in Food and Cosmetics Toxicology. Elsevier. ISBN 978-1-4831-4797-0.
- ^ Mattiello, Joseph J. (1945). Protective and Decorative Coatings. U.S. Government Printing Office.
- ^ Opdyke, D. L. J. (2013-10-22). Monographs on Fragrance Raw Materials: A Collection of Monographs Originally Appearing in Food and Cosmetics Toxicology. Elsevier. ISBN 978-1-4831-4797-0.



