Vaginal cancer
| Vaginal cancer | |
|---|---|
| Specialty | Gynecologic Oncology |
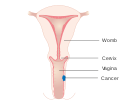
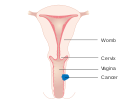
Vaginal cancer is an extraordinarily rare form of cancer that develops in the tissue of the vagina.[1] Primary vaginal cancer originates from the vaginal tissue – most frequently squamous cell carcinoma, but primary vaginal adenocarcinoma, sarcoma, and melanoma have also been reported[2] – while secondary vaginal cancer involves the metastasis of a cancer that originated in a different part of the body. Secondary vaginal cancer is more common.[3] Signs of vaginal cancer may include abnormal vaginal bleeding, dysuria, tenesmus, or pelvic pain,[4][5] though as many as 20% of women diagnosed with vaginal cancer are asymptomatic at the time of diagnosis.[6] Vaginal cancer occurs more frequently in women over age 50, and the mean age of diagnosis of vaginal cancer is 60 years.[7] It often can be cured if found and treated in early stages. Surgery alone or surgery combined with pelvic radiation is typically used to treat vaginal cancer.
Description
[edit]Carcinoma of the vagina occurs in less than 2% of women with pelvic malignant tumors. Squamous carcinoma is the most common type of vaginal cancer. The human papilloma virus (HPV) is strongly associated with vaginal cancer. Vaginal cancer occurs most often in the upper third of the vagina (51%), 30% are found in the lower third, and 19% in the middle third. Vaginal cancer can present as an elevated lesion growing out from the epithelial surface or an ulcer-like, shallow depression. Definitive diagnosis is determined by biopsy.[8]
Signs and symptoms
[edit]Most vaginal cancers do not cause signs or symptoms early on. When vaginal cancer does cause symptoms, they may include:
- Vaginal discharge or abnormal bleeding
- Unusually heavy flow of blood
- Bleeding after menopause
- Bleeding between periods; or any other
- Bleeding that is longer than normal
- Blood in the stool or urine
- Frequent or urgent need to urinate
- Feeling constipated[9]
- Pain during sexual intercourse
- A lump or growth in the vagina that can be felt.[10]
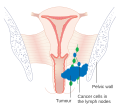
Enlarged pelvic lymph nodes can sometimes be palpated.[11]
Risk factors
[edit]- Prenatal exposure to diethylstilbestrol
- Infection with human papillomavirus (HPV) type 16
- Infection with human immunodeficiency virus (HIV) type 1[12][13]
- Previous history of cervical cancer
- Smoking
- Chronic vulvar itching or burning[9]
Types
[edit]There are two primary types of vaginal cancer: squamous-cell carcinoma and adenocarcinoma.[14]
- Squamous-cell carcinoma of the vagina arises from the squamous cells (epithelium) that line the vagina. This is the most common type of vaginal cancer. It is found most often in women aged 60 or older.
- Vaginal adenocarcinoma arises from the glandular (secretory) cells in the lining of the vagina. Adenocarcinoma is more likely to spread to the lungs and lymph nodes.
- Clear cell adenocarcinoma occurs in a small percentage of women (termed "DES-Daughters") born between 1938 and 1973 (later outside the United States) that were exposed to the drug diethylstilbestrol (DES) in utero. DES was prescribed to 5 to 10 million mothers period to prevent possible miscarriages and premature births.[15] Typically, women develop DES-related adenocarcinoma before age 30, but increasing evidence suggests possible effects or cancers (including other forms of vaginal glandular tumors) at a later age. DES-exposure in women is also linked to various infertility and pregnancy complications. Daughters exposed to DES in utero may also have an increased risk of moderate/severe cervical squamous cell dysplasia and an increased risk of breast cancer.[15] Approximately one in 1,000 (0.1%) DES daughters will be diagnosed with clear cell adenocarcinoma. The risk is virtually non-existent among premenopausal women not exposed to DES.[16]
- Vaginal germ cell tumors (primarily teratoma and endodermal sinus tumor) are rare. They are found most often in infants and children.
- Sarcoma botryoides, a rhabdomyosarcoma also is found most often in infants and children.
- Vaginal melanoma, a melanoma that appears in the vagina.
-
Stage 1 vaginal cancer
-
Stage 2 vaginal cancer
-
Stage 3 vaginal cancer
-
Stage 4A vaginal cancer
-
Stage 4B vaginal cancer
Diagnosis and Screening
[edit]Routine vaginal cancer screening, including routine surveillance imaging such as ultrasound or MRI, is not recommended for women who do not have symptoms. Imaging without indications is discouraged because it is unlikely to detect a recurrence or improve survival, and because it has its own costs and side effects.[17][18] Several tests are used to diagnose vaginal cancer, including:
- Physical exam and history
- Pelvic exam
- Biopsy
- Colposcopy
MRI provides visualization of the extent of vaginal cancer.[19] Other sources of cancer tissue such as the urethra or the cervix must be ruled out before a diagnosis of vaginal cancer is made.[20] Vaginal cancer cannot be detected through cervical pap smears.[9]
Classification
[edit]Staging: The International Federation of Gynecology and Obstetrics utilizes the Tumor, Node, Metastasis (TNM) method of staging vaginal cancer. The clinical staging of the most common form of primary vaginal cancer, squamous cell carcinoma, is outlined below.[21]
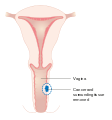
- Stage I - Tumor confined to the vagina
- Stage II - Tumor invading tissue near the vagina, but has not spread to pelvic wall
- Stage III- Tumor invading pelvic sidewall or interfering with renal function, obstructing ureter to cause hydronephrosis.
- Stage IV - Tumor invading mucosa of nearby pelvic organs, including bladder or rectum, or has spread beyond pelvis.
Management
[edit]Historically, the combination of external-beam radiation therapy (EBRT) has been the most common treatment for vaginal cancer. In early stages of vaginal cancer, surgery also has some benefit. This management and treatment is less effective for those with advanced stages of cancer but works well in early stages with high rates of cure. Advanced vaginal cancer only has a 5-year survival rates of 52.2%, 42.5% and 20.5% for patients with stage II, III and IVa disease. Newer treatments for advanced stages of ovarian have been developed. These utilize concurrent carboplatin plus paclitaxel, EBRT and high-dose-rate interstitial brachytherapy (HDR-ISBT).[11]
When the chance of surgical removal of all cancerous tissue is very low or when the surgery has a chance of damaging the bladder, vagina or bowel, radiation therapy is used. When a tumor is less than 4 cm in diameter, radiation therapy provides excellent results. In these instances, the 5-year survival rate is greater than 80%.[11] Treatments are individualized due to the rarity of vaginal cancer studies.[22]
-
A local surgery to remove vaginal cancer
-
A radical hysterectomy to treat vaginal cancer without reconstruction
-
A radical hysterectomy for vaginal cancer with reconstruction of the vagina using other tissues
Epidemiology
[edit]Cancer of the vagina is rare and is only 2% of all gynecological cancers and less than 0.5% of all cancers in women.[11] Estimated new cases in the United States in 2017 are 4,810. Deaths from vaginal cancer during the same time were 1,240.[14] It is more common in older women.[23]
In the UK, 254 cases of vaginal cancer were identified in 2014. Deaths from vaginal cancer in this period were 110.[24] Out of those with vaginal cancer, 53% are related to HPV infection.[12]
Research
[edit]Clinical trials
[edit]- Because vaginal cancer is so rare, there have been very few phase three clinical trials studying treatments specifically targeting vaginal cancer.[25] There is an ongoing phase three trial studying the efficacy of the drug triapine to treat vaginal cancer.[26] There are currently several phase one and two clinical trials underway to study the efficacy of new gynecological cancer treatment regimens, which include treatment for vaginal cancer.[27][28]
See also
[edit]References
[edit]- ^ Siegel, Rebecca L.; Miller, Kimberly D.; Fuchs, Hannah E.; Jemal, Ahmedin (January 2021). "Cancer Statistics, 2021". CA: A Cancer Journal for Clinicians. 71 (1): 7–33. doi:10.3322/caac.21654. ISSN 1542-4863. PMID 33433946.
- ^ Berek, JS (2015). Berek and Hacker's Gynecologic Oncology, 6th ed. Philadelphia: Lippincott Williams & Wilkins. p. 608. ISBN 9781451190076.
- ^ Dunn, Leo J.; Napier, John G. (1966-12-15). "Primary carcinoma of the vagina". American Journal of Obstetrics and Gynecology. 96 (8): 1112–1116. doi:10.1016/0002-9378(66)90519-9. ISSN 0002-9378. PMID 5928467.
- ^ Choo, Y. C.; Anderson, D. G. (August 1982). "Neoplasms of the vagina following cervical carcinoma". Gynecologic Oncology. 14 (1): 125–132. doi:10.1016/0090-8258(82)90059-2. hdl:2027.42/23906. ISSN 0090-8258. PMID 7095583.
- ^ Herbst, A. L.; Ulfelder, H.; Poskanzer, D. C. (1971-04-15). "Adenocarcinoma of the vagina. Association of maternal stilbestrol therapy with tumor appearance in young women". The New England Journal of Medicine. 284 (15): 878–881. doi:10.1056/NEJM197104222841604. ISSN 0028-4793. PMID 5549830.
- ^ Underwood, P. B.; Smith, R. T. (1971-07-05). "Carcinoma of the vagina". JAMA. 217 (1): 46–52. doi:10.1001/jama.1971.03190010028006. ISSN 0098-7484. PMID 4932433.
- ^ Creasman, W. T.; Phillips, J. L.; Menck, H. R. (1998-09-01). "The National Cancer Data Base report on cancer of the vagina". Cancer. 83 (5): 1033–1040. doi:10.1002/(SICI)1097-0142(19980901)83:5<1033::AID-CNCR30>3.0.CO;2-6. ISSN 0008-543X. PMID 9731908.
- ^ Shobeiri, S. Abbas; Rostaminia, Ghazaleh; White, Dena; Quiroz, Lieschen H.; Nihira, Mikio A. (2013-08-01). "Evaluation of Vaginal Cysts and Masses by 3-Dimensional Endovaginal and Endoanal Sonography". Journal of Ultrasound in Medicine. 32 (8): 1499–1507. doi:10.7863/ultra.32.8.1499. ISSN 1550-9613. PMID 23887963.
- ^ a b c "Vaginal and Vulvar Cancer" (PDF). Centers for Disease Control and Prevention. December 2016. Retrieved 18 December 2017.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "Symptoms | Vaginal cancer | Cancer Research UK". www.cancerresearchuk.org. 10 September 2015. Retrieved 2017-12-19.
- ^ a b c d Mabuchi, Seiji; Kawano, Mahiru; Isohashi, Fumiaki; Kuroda, Hiromasa; Sasano, Tomoyuki; Kimura, Tadashi (2015-01-01). "First two cases of primary carcinoma of the vagina successfully treated with concurrent weekly carboplatin plus paclitaxel, external beam radiotherapy and high-dose-rate interstitial brachytherapy: A case report and published work review". Journal of Obstetrics and Gynaecology Research. 41 (1): 156–161. doi:10.1111/jog.12492. ISSN 1447-0756. PMID 25227150. S2CID 23908930.
- ^ a b "Vaginal cancer risk factors". Cancer Research UK. 2015-05-15. Retrieved 2017-12-13.
- ^ "List of Classifications by cancer sites with sufficient or limited evidence in humans, Volumes 1 to 120 a". World Health Organization, International Agency for Research on Cancer. 2017. Retrieved 13 December 2017.
- ^ a b "Vaginal Cancer Treatment". National Institutes of Health, National Cancer Institute. 9 February 2017. Retrieved 2017-12-13.
- ^ a b "About DES". Centers for Disease Control and Prevention. Retrieved 2017-12-13.
- ^ "Known Health Effects for DES Daughters". Centers for Disease Control and Prevention. Retrieved 2017-12-13.
- ^ Society of Gynecologic Oncology (February 2014), "Five Things Physicians and Patients Should Question", Choosing Wisely: an initiative of the ABIM Foundation, Society of Gynecologic Oncology, retrieved 19 February 2013
- ^ "What Should I Know About Screening for Vaginal and Vulvar Cancers? | CDC". www.cdc.gov. 2021-08-23. Retrieved 2021-09-13.
- ^ Halperin, Edward C.; Brady, Luther W.; Perez, Carlos A.; Wazer, David E. (2013-05-06). Perez and Brady's principles and practice of radiation oncology. Halperin, Edward C.,, Brady, Luther W., 1925-, Wazer, David E.,, Perez, Carlos A., 1934- (Sixth ed.). Philadelphia. p. 1412. ISBN 9781451116489. OCLC 849739571.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Khan, Michelle J.; Massad, L. Stewart; Kinney, Walter; Gold, Michael A.; Mayeaux, E. J.; Darragh, Teresa M.; Castle, Philip E.; Chelmow, David; Lawson, Herschel W.; Huh, Warner K. (April 2016). "A Common Clinical Dilemma: Management of Abnormal Vaginal Cytology and Human Papillomavirus Test Results". Journal of Lower Genital Tract Disease. 20 (2): 119–125. doi:10.1097/LGT.0000000000000185. ISSN 1526-0976. PMID 26901279. S2CID 10644537.
- ^ Shah, Chirag A.; Goff, Barbara A.; Lowe, Kimberly; Peters, William A.; Li, Christopher I. (May 2009). "Factors affecting risk of mortality in women with vaginal cancer". Obstetrics and Gynecology. 113 (5): 1038–1045. doi:10.1097/AOG.0b013e31819fe844. ISSN 0029-7844. PMC 2746762. PMID 19384118.
- ^ Damast, Shari; Takiar, Vinita; McCarthy, Shirley; Higgins, Susan A. (2016). "Treatment of early stage vaginal cancer with EBRT and MRI-based intracavitary brachytherapy: A retrospective case review". Gynecologic Oncology Reports. 17: 89–92. doi:10.1016/j.gore.2016.08.002. PMC 4975702. PMID 27536721.
- ^ "Vaginal cancer | Vaginal cancer | Cancer Research UK". www.cancerresearchuk.org. Retrieved 2017-12-19.
- ^ "Vaginal cancer statistics". Cancer Research UK. 2015-05-14. Retrieved 2017-12-13.
- ^ Di Donato, Violante; Bellati, Filippo; Fischetti, Margherita; Plotti, Francesco; Perniola, Giorgia; Panici, Pierluigi Benedetti (March 2012). "Vaginal cancer". Critical Reviews in Oncology/Hematology. 81 (3): 286–295. doi:10.1016/j.critrevonc.2011.04.004. ISSN 1879-0461. PMID 21571543.
- ^ "A Study of Combination Radiation Therapy and Cisplatin with or without Triapine for Treating Patients with Newly Diagnosed Stage IB2, II, or IIIB-IVA Cervical Cancer or Stage II-IVA Vaginal Cancer". Mayo Clinic. Retrieved 2021-09-13.
- ^ M.D. Anderson Cancer Center (2020-12-01). "A Phase 2, Open-Label Study to Evaluate Efficacy of Combination Treatment With MEDI0457 (INO-3112) and Durvalumab (MEDI4736) in Patients With Recurrent/Metastatic Human Papilloma Virus Associated Cancers". National Cancer Institute (NCI).
- ^ Naumann, R. Wendel; Hollebecque, Antoine; Meyer, Tim; Devlin, Michael-John; Oaknin, Ana; Kerger, Joseph; López-Picazo, Jose M.; Machiels, Jean-Pascal; Delord, Jean-Pierre; Evans, Thomas R. J.; Boni, Valentina (2019-11-01). "Safety and Efficacy of Nivolumab Monotherapy in Recurrent or Metastatic Cervical, Vaginal, or Vulvar Carcinoma: Results From the Phase I/II CheckMate 358 Trial". Journal of Clinical Oncology. 37 (31): 2825–2834. doi:10.1200/JCO.19.00739. ISSN 1527-7755. PMC 6823884. PMID 31487218.