Tetrahydrofolic acid

| |

| |
| Names | |
|---|---|
| IUPAC name
N-[4-({[(6Ξ)-2-Amino-4-oxo-1,4,5,6,7,8-hexahydropteridin-6-yl]methyl}amino)benzoyl]-L-glutamic acid
| |
| Systematic IUPAC name
(2S)-2-[4-({[(6Ξ)-2-Amino-4-oxo-1,4,5,6,7,8-hexahydropteridin-6-yl]methyl}amino)benzamido]pentanedioic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| 101189 | |
| ChEBI | |
| ChemSpider | |
| DrugBank | |
| KEGG | |
| MeSH | 5,6,7,8-tetrahydrofolic+acid |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C19H23N7O6 | |
| Molar mass | 445.43 g/mol |
| Melting point | 250 °C (482 °F; 523 K) |
| 0.27 g/L | |
| Acidity (pKa) | 3.51 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tetrahydrofolic acid (THFA), or tetrahydrofolate, is a folic acid derivative.
Metabolism
[edit]Human synthesis
[edit]Tetrahydrofolic acid is produced from dihydrofolic acid by dihydrofolate reductase. This reaction is inhibited by methotrexate.[1]
It is converted into 5,10-methylenetetrahydrofolate by serine hydroxymethyltransferase.
Bacterial synthesis
[edit]Many bacteria use dihydropteroate synthetase to produce dihydropteroate, a molecule without function in humans. This makes it a useful target for sulfonamide antibiotics, which compete with the PABA precursor.
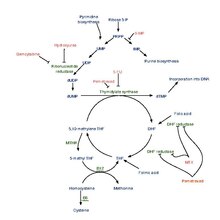
Functions
[edit]Tetrahydrofolic acid is a cofactor in many reactions, especially in the synthesis (or anabolism) of amino acids and nucleic acids. In addition, it serves as a carrier molecule for single-carbon moieties, that is, groups containing one carbon atom e.g. methyl, methylene, methenyl, formyl, or formimino. When combined with one such single-carbon moiety as in 10-formyltetrahydrofolate, it acts as a donor of a group with one carbon atom. Tetrahydrofolate gets this extra carbon atom by sequestering formaldehyde produced in other processes. These single-carbon moieties are important in the formation of precursors for DNA synthesis. A shortage in tetrahydrofolic acid (FH4) can cause megaloblastic anemia.[2][3][4]
Methotrexate acts on dihydrofolate reductase, like pyrimethamine or trimethoprim, as an inhibitor and thus reduces the amount of tetrahydrofolate made. This may result in megaloblastic anemia.
Tetrahydrofolic acid is involved in the conversion of formiminoglutamic acid to glutamic acid; this may reduce the amount of histidine available for decarboxylation and protein synthesis, and hence the urinary histamine and formiminoglutamic acid may be decreased.[5]
References
[edit]- ^ Rajagopalan, P. T. Ravi; Zhang, Zhiquan; McCourt, Lynn; Dwyer, Mary; Benkovic, Stephen J.; Hammes, Gordon G. (2002-10-15). "Interaction of dihydrofolate reductase with methotrexate: Ensemble and single-molecule kinetics". Proceedings of the National Academy of Sciences. 99 (21): 13481–13486. Bibcode:2002PNAS...9913481R. doi:10.1073/pnas.172501499. ISSN 0027-8424. PMC 129699. PMID 12359872.
- ^ "Biochemistry: The One-Carbon Pool: Folate and B12 Metabolism". liveonearth.livejournal.com. 2008-02-23. Retrieved 2020-12-15.
- ^ Yadav, Manish K.; Manoli, Nandini M.; Madhunapantula, SubbaRao V. (2016-10-25). Roemer, Klaus (ed.). "Comparative Assessment of Vitamin-B12, Folic Acid and Homocysteine Levels in Relation to p53 Expression in Megaloblastic Anemia". PLOS ONE. 11 (10): e0164559. Bibcode:2016PLoSO..1164559Y. doi:10.1371/journal.pone.0164559. ISSN 1932-6203. PMC 5079580. PMID 27780269.
- ^ Aslinia, F.; Mazza, J. J.; Yale, S. H. (2006-09-01). "Megaloblastic Anemia and Other Causes of Macrocytosis". Clinical Medicine & Research. 4 (3): 236–241. doi:10.3121/cmr.4.3.236. ISSN 1539-4182. PMC 1570488. PMID 16988104.
- ^ Dawson W, Maudsley DV, West GB (December 1965). "Histamine formation in guinea-pigs". J. Physiol. 181 (4): 801–9. doi:10.1113/jphysiol.1965.sp007798. PMC 1357684. PMID 5881255.


