Parvovirus B19
| Primate erythroparvovirus 1 | |
|---|---|
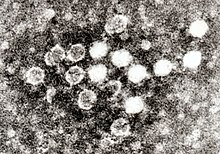
| |
| Electron micrograph of Parvoviruses in blood | |
| Virus classification | |
| (unranked): | Virus |
| Realm: | Monodnaviria |
| Kingdom: | Shotokuvirae |
| Phylum: | Cossaviricota |
| Class: | Quintoviricetes |
| Order: | Piccovirales |
| Family: | Parvoviridae |
| Genus: | Erythroparvovirus |
| Species: | Primate erythroparvovirus 1
|
| Synonyms | |
| |
Human parvovirus B19, generally referred to as B19 virus (B19V), parvovirus B19[1] or sometimes erythrovirus B19,[2] is a known human virus in the family Parvoviridae, genus Erythroparvovirus; it measures only 23–26 nm in diameter.[3] Human parvovirus b19 is a below-species classification of Erythroparvovirus primate1.[4] The name is derived from Latin parvum, meaning small, reflecting the fact that B19 ranks among the smallest DNA viruses. B19 virus is most known for causing disease in the pediatric population; however, it can also affect adults. It is the classic cause of the childhood rash called fifth disease or erythema infectiosum, or "slapped face syndrome".[5][6] The name comes from it being the fifth in a list of historical classifications of common skin rash illnesses in children.[7]
The virus was discovered by chance in 1975 by Australian virologist Yvonne Cossart.[3][6] The name B19 originated from the coding of a serum sample, number 19 in panel B.[6][8]
Virology
[edit]Erythroviruses belong to the Parvoviridae family of small DNA viruses.[9] Human parvovirus B19 is a non-enveloped, icosahedral virus that contains a single-stranded linear DNA genome of approximately 5,600 base pairs in length.[10] B19V's nonenveloped viral particles are ~22 to 24 nm in diameter which are quite small compared to other parvoviruses.[3] The infectious particles may contain either positive or negative strands of DNA. The icosahedral capsid consists of 60 capsomeres, consisting of two structural proteins, VP1 (83 kDa) and VP2 (58 kDa), which are identical except for 227 amino acids at the amino-terminal of the VP1-protein, the so-called VP1-unique region. VP2 is the major capsid protein, and comprises approximately 95% of the total virus particle. VP1-proteins are incorporated into the capsid structure in a non-stoichiometrical relation (based on antibody-binding analysis and X-ray structural analysis the VP1-unique (VP1u) region is assumed to be exposed at the surface of the virus particle.[11] VP1u is located in the N-terminus of the VP1.[12] At each end of the DNA molecule there are palindromic sequences which form "hairpin" loops. The hairpin at the 3' end serves as a primer for the DNA polymerase.[13] It is classified as an erythrovirus because of its capability to invade red blood cell precursors in the bone marrow. Three genotypes (with subtypes) have been recognised.[14]
The genome of human parvovirus B19 encodes four other proteins in addition to VP1 and VP2 and also some smaller proteins whose function is still unknown.[15] The most notable of these is the large nonstructural protein commonly referred to as NS1. NS1 is a multifunctional protein that has a role in binding and operating on the p6 promoter which gives NS1 the ability to control the transcription of the B19V genome.[16] NS1 sequence-specifically binds and cleaves DNA via restriction endonuclease activity at its N-terminus.[17] NS1 is responsible for the regulation of certain cellular promoters including the p21/WAF1 promoter,[18] and is thought to regulate the virus' own promoter.[19] The 11 kDa protein encoded by the viral genome has been implicated in viral DNA replication.[20]
The nucleotide substitution rate for total coding DNA has been estimated to be 1.03 (0.6-1.27) x 10−4 substitutions/site/year.[21] This rate is similar to that of other single-stranded DNA viruses. VP2 codons were found to be under purifying selection. In contrast VP1 codons in the unique part of the gene were found to be under diversifying selection. This diversifying selection is consistent with persistent infection as this part of the VP1 protein contains epitopes recognised by the immune system.[citation needed]
Like other nonenveloped DNA viruses, pathogenicity of parvovirus B19 involves binding to host cell receptors, internalization, translocation of the genome to the host nucleus, DNA replication, RNA transcription, assembly of capsids and packaging of the genome, and finally cell lysis with release of the mature virions.[22] In humans the P antigen (also known as globoside) is the cellular receptor for parvovirus B19 virus that causes erythema infectiosum (fifth disease) in children. This infection is sometimes complicated by severe aplastic anemia caused by lysis of early erythroid precursors.[citation needed]
Studies of Parvovirus B19 (B19V) have provided insights into its persistence and integration within human tissues. Analyses revealed that B19V primarily resides in endothelial cells, monocytes, and B cells, with few documented integration events in vivo. Although B19V DNA integration has been observed in vitro, its low prevalence in sampled tissues suggests that alternative mechanisms may contribute to lifelong persistence. In one case involving pulmonary carcinoma, a B19V integration junction was found in the colon, indicating possible interactions with host genomic elements.[23]
Evolution
[edit]The most recent common ancestor of the extant strains has been dated to about 12,600 years ago.[24] Three genotypes—1, 2 and 3—are recognised. A recombination between types 1 and 3 gave rise to genotype 2 between 5,000 and 6,800 years ago.[citation needed]
Transmission
[edit]The virus is primarily spread by infected respiratory droplets; however, blood-borne transmission has also been reported.[25] The secondary attack risk for exposed household persons is about 50%, and about half of that for classroom contacts.[6][26] Transmission can happen from a mother that has B19V infection during pregnancy. The baby can get infected by bloodstream and therefore develop a severe anemia.[27]
Infectivity and symptoms
[edit]Two out of ten infected people do not experience any symptoms but are still highly infective. The B19V infection starts with flu-like symptoms consisting fever, headache, runny nose, sore throat, joint pain and rash.[28] Symptoms begin some six days after exposure (between 4 and 28 days, with the average being 16 to 17 days[29]) and last about a week. The rash that children experience will appear after few days after the initial symptoms and can spread all over the body.[28]
Infected patients with normal immune systems are contagious before becoming symptomatic, but probably not after.[30] Individuals with B19 IgG antibodies are generally considered immune to recurrent infection, but reinfection is possible in a minority of cases.[31] About half of adults are B19-immune due to a past infection.[27]
Immunocompromised patients (organ transplant, HIV etc.) are prone to complications that affect the nerves, joints or bloodstream.[28]
Epidemiology
[edit]A significant increase in the number of cases is seen every three to four years; the last epidemic year was 1998.[32] Outbreaks can arise especially in nurseries and schools.
Parvovirus B19 causes an infection in humans only. Cat and dog parvoviruses do not infect humans due to animals having their own parvoviruses. There is always a possibility for a spillover. There is no vaccine available for human parvovirus B19,[33] though attempts have been made to develop one.[34][35]
Role in disease
[edit]

Fifth disease
[edit]Fifth disease or erythema infectiosum is only one of several expressions of parvovirus B19. The associated bright red rash of the cheeks gives it the nickname "slapped cheek syndrome".[6] Any age may be affected, although it is most common in children aged six to ten years. It is so named because it was the fifth most common cause of a pink-red infection associated rash to be described by physicians (many of the others, such as measles and rubella, are rare now).[36]
Once infected, patients usually develop the illness after an incubation period of four to fourteen days. The disease commences with high fever and malaise, when the virus is most abundant in the bloodstream, and patients are usually no longer infectious once the characteristic rash of this disease has appeared.[33] The following symptoms are characteristic:
- A usual brief viral prodrome with fever, headache, nausea, diarrhea.
- As the fever breaks, a red rash forms on the cheeks, with relative pallor around the mouth ("slapped cheek rash"), sparing the nasolabial folds, forehead, and mouth.
- "Lace-like, (reticular)" red rash on trunk or extremities then follows the facial rash. Infection in adults usually only involves the reticular rash, with multiple joint pain predominating.
- Exacerbation of rash by sunlight, heat, stress.
Papular purpuric gloves and socks syndrome
[edit]Teenagers or young adults may develop the so-called Papular purpuric gloves and socks syndrome. It is a cutaneous condition characterized by pruritus, edema, and erythema of the hands and feet.[37]: 401 In 1996, an association with parvovirus B19 was described, after virus was demonstrated in skin biopsy samples,[38] subsequently corroborated in numerous publications.[39] [40]
Polyarthropathy syndrome
[edit]Arthralgias and arthritis are commonly reported in association with parvovirus B19 infection in adults in the absence of skin findings, whereas erythema infectiosum is the main symptom observed in children. The occurrence of arthralgia coincides with the initial detection of circulating IgM- and IgG-antibodies against the viral structural proteins VP1 and VP2. Parvovirus B19 infection may affect the development of arthritis.[11] In adults (and perhaps some children), parvovirus B19 can lead to a seronegative arthritis which is usually easily controlled with analgesics.[41] Women are approximately twice as likely as men to experience arthritis after parvovirus infection. Possibly up to 15% of all new cases of arthritis are due to parvovirus, and a history of recent contact with a patient and positive serology generally confirms the diagnosis.[30] This arthritis does not progress to other forms of arthritis. Typically joint symptoms last 1–3 weeks, but in 10–20% of those affected, it may last weeks to months.[6][33]
A Danish study has links between B19 with polymyalgia rheumatica.[42]
Aplastic crisis and chronic erythroid hypoplasia
[edit]Although most patients have a decrease of erythropoiesis (production of red blood cells) during parvovirus infection, it is most dangerous in patients with pre-existing bone marrow stress, for example sickle cell anemia or hereditary spherocytosis,[43][44] and are therefore heavily dependent on erythropoiesis due to the reduced lifespan of the red cells. This is termed "aplastic crisis" (also called reticulocytopenia). It is treated with blood transfusion.
Parvovirus B19 is a cause of chronic anemia in individuals with immunodeficiency, receiving immunosuppressive therapy or with HIV infection. Treatment with intravenous immunoglobulin usually resolves the anemia although relapse can occur. Parvovirus infection may trigger an inflammatory reaction in AIDS patients who have just begun antiretroviral therapy.[45]
Hydrops fetalis
[edit]
Parvovirus infection in pregnant women is associated with hydrops fetalis due to severe fetal anemia, sometimes leading to miscarriage or stillbirth.[33][46] This is due to a combination of hemolysis of the red blood cells, as well as the virus directly negatively affecting the red blood cell precursors in the bone marrow. The risk of fetal loss is about 10% if infection occurs before pregnancy week 20 (especially between weeks 14 and 20), but minimal after then. Routine screening of the antenatal sample would enable the pregnant mother to determine the risk of infection.[citation needed] Knowledge of her status would allow the mother to avoid contact with individuals suspected or known to have an ongoing infection, however, at the present time, antenatal testing for immunity is not recommended, since there is no good means to prevent the infection, there is no specific therapy and there are no vaccines available. It may increase maternal anxiety and fear without proven benefit. The best approach would be to recommend all pregnant women to avoid contact with children with current symptoms of infection, as described above. The risk to the fetus will be reduced with correct diagnosis of the anemia (by ultrasound scans) and treatment (by blood transfusions). There is some evidence that intrauterine parvovirus B19 infection leads to developmental abnormalities in childhood.[47]
Treatment
[edit]At the moment, there are no treatments that directly target parvovirus B19 virus.[48] Intravenous immunoglobulin therapy (IVIG) therapy has been a popular alternative because doctors can administer it without stopping chemotherapy drugs like MEL-ASCT.[49] Also, the treatment's side effects are rare as only 4 out of 133 patients had complications (2 had acute kidney injury and 2 had pulmonary edema) even though 69 of the patients had organ transplants and 39 of them were HIV positive.[50] This is a large improvement over administering rituximab. [citation needed] The monoclonal antibody against the CD20 protein has been shown to cause acute hepatitis,[51] neutropenia via parvovirus B19 reactivations,[52] and even persistent parvovirus B19 infection.[53] However, it is important to note that IVIG therapy is not perfect as 34% of treated patients will have a relapse after 4 months.[50]
Diagnostics and Vaccination
[edit]There is no routine laboratory test for B19V, but in case of the doctor is suspecting a B19V infection patient's blood will be drawn. The blood is then tested for antibodies.[27]
As of 2020, no approved human vaccine existed against parvovirus B19.[54] Bernstein et. al.[35] tested in 2011 a possible candidate vaccine that was produced in insect cells. For the vaccine VP2/VP1 VLPs (virus-like particle) were used. This vaccine was approved to the I clinical phase, but was stopped due to unexplained reactions in patients. It was argued that the reaction could have been due to the PLA2 activity in VP1u. The reaction was either to the PLA2 activity releasing arachidonic acid and precursors of inflammatory mediators or the usage of insect cells. The PLA2 activity can be reduced or fully removed with site-directed mutagenesis. So this could still act as a candidate vaccine, but further research is needed.[12]
See also
[edit]References
[edit]- ^ Servey JT, Reamy BV, Hodge J (February 2007). "Clinical presentations of parvovirus B19 infection". American Family Physician. 75 (3): 373–376. PMID 17304869.
- ^ Kahn JS, Kesebir D, Cotmore SF, D'Abramo A, Cosby C, Weibel C, Tattersall P (July 2008). "Seroepidemiology of human bocavirus defined using recombinant virus-like particles". The Journal of Infectious Diseases. 198 (1): 41–50. doi:10.1086/588674. PMID 18491974.
- ^ a b c Heegaard ED, Brown KE (July 2002). "Human parvovirus B19". Clinical Microbiology Reviews. 15 (3): 485–505. doi:10.1128/CMR.15.3.485-505.2002. PMC 118081. PMID 12097253.
- ^ "Human parvovirus B19". NCBI. Retrieved 7 January 2024.
- ^ Vafaie J, Schwartz RA (October 2004). "Parvovirus B19 infections". International Journal of Dermatology. 43 (10): 747–749. doi:10.1111/j.1365-4632.2004.02413.x. PMID 15485533. S2CID 25660330.
- ^ a b c d e f Sabella C, Goldfarb J (October 1999). "Parvovirus B19 infections". American Family Physician. 60 (5): 1455–1460. PMID 10524489. Archived from the original on 14 May 2008. Retrieved 6 November 2009.
- ^ "Parvovirus B19". CDC. Retrieved 25 February 2024.
- ^ Siegl, Günter; Bates, Robert C.; Berns, Kenneth I.; Carter, Barrie J.; Kelly, David C.; Kurstak, Edouard; Tattersall, Peter (25 July 2008). "Characteristics and Taxonomy of Parvoviridae". Intervirology. 23 (2): 61–73. doi:10.1159/000149587. ISSN 0300-5526. PMID 3980186.
- ^ Brown KE (2004). "Variants of B19". Developments in Biologicals. 118: 71–77. PMID 15645675.
- ^ Luo Y, Qiu J (2015). "Human parvovirus B19: a mechanistic overview of infection and DNA replication". Future Virology. 10 (2): 155–167. doi:10.2217/fvl.14.103. PMC 4470565. PMID 26097496.
- ^ a b von Landenberg P, Lehmann HW, Modrow S (April 2007). "Human parvovirus B19 infection and antiphospholipid antibodies". Autoimmunity Reviews. 6 (5): 278–85. doi:10.1016/j.autrev.2006.09.006. PMID 17412298.
- ^ a b Soto-Valerio, I. A.; Cayetano-Cruz, M.; Valadez-García, J.; Guadarrama, P.; Méndez, C.; Bustos-Jaimes, I. (2022). "In vitro refolding of the structural protein VP1 of parvovirus B19 produces virus-like particles with functional VP1 unique region". Virology. 570: 57–66. doi:10.1016/j.virol.2022.03.009. PMID 35367742. Retrieved 25 February 2024.
- ^ G. Siegl and P. Cassinotti, Parvoviruses Chapter 14, Topley and Wison's Microbiology and Microbial Infections, Vol. 1, Virology, 1998 pp. 261–280
- ^ Molenaar-de Backer MW, Lukashov VV, van Binnendijk RS, Boot HJ, Zaaijer HL (2012). "Global co-existence of two evolutionary lineages of parvovirus B19 1a, different in genome-wide synonymous positions". PLOS ONE. 7 (8): e43206. Bibcode:2012PLoSO...743206M. doi:10.1371/journal.pone.0043206. PMC 3418230. PMID 22912828.
- ^ Gallinella G (April 2013). "Parvovirus B19 Achievements and Challenges". ISRN Virology. 2013: e898730. doi:10.5402/2013/898730. S2CID 84592174.
- ^ Zhang, Y.; Shao, Z.; Gao, Y.; Fan, B.; Yang, J.; Chen, X.; Zhao, X.; Shao, Q.; Zhang, W.; Cao, C.; Liu, H.; Gan, J. (2022). "Structures and implications of the nuclease domain of human parvovirus B19 NS1 protein". Computational and Structural Biotechnology Journal. 20: 4645–4655. doi:10.1016/j.csbj.2022.08.047. PMC 9440244. PMID 36090819.
- ^ Sanchez JL, Romero Z, Quinones A, Torgeson KR, Horton NC (November 2016). "DNA Binding and Cleavage by the Human Parvovirus B19 NS1 Nuclease Domain". Biochemistry. 55 (47): 6577–6593. doi:10.1021/acs.biochem.6b00534. PMC 5933076. PMID 27809499.
- ^ Nakashima A, Morita E, Saito S, Sugamura K (November 2004). "Human Parvovirus B19 nonstructural protein transactivates the p21/WAF1 through Sp1". Virology. 329 (2): 493–504. doi:10.1016/j.virol.2004.09.008. PMID 15518826.
- ^ Raab U, Beckenlehner K, Lowin T, Niller HH, Doyle S, Modrow S (February 2002). "NS1 protein of parvovirus B19 interacts directly with DNA sequences of the p6 promoter and with the cellular transcription factors Sp1/Sp3". Virology. 293 (1): 86–93. doi:10.1006/viro.2001.1285. PMID 11853402.
- ^ Xu P, Chen AY, Ganaie SS, Cheng F, Shen W, Wang X, et al. (January 2019). Jung JU (ed.). "The 11-Kilodalton Nonstructural Protein of Human Parvovirus B19 Facilitates Viral DNA Replication by Interacting with Grb2 through Its Proline-Rich Motifs". Journal of Virology. 93 (1). doi:10.1128/JVI.01464-18. PMC 6288338. PMID 30282717.
- ^ Stamenković GG, Ćirković VS, Šiljić MM, Blagojević JV, Knežević AM, Joksić ID, Stanojević MP (October 2016). "Substitution rate and natural selection in parvovirus B19". Scientific Reports. 6: 35759. Bibcode:2016NatSR...635759S. doi:10.1038/srep35759. PMC 5075947. PMID 27775080.
- ^ Aslanidis et al. Parvovirus B19 infection and systemic lupus erythematosus: Activation of an aberrant pathway?, 2007.
- ^ Pyöriä L, Pratas D, Toppinen M, Simmonds P, Hedman K, Sajantila A, Perdomo M (2024). "Intra-host genomic diversity and integration landscape of human tissue-resident DNA virome". Nucleic Acids Research. 52 (21): gkae871. doi:10.1093/nar/gkae871. PMC 11602146. PMID 39436041.
- ^ Mühlemann B, Margaryan A, Damgaard PB, Allentoft ME, Vinner L, Hansen AJ, et al. (July 2018). "Ancient human parvovirus B19 in Eurasia reveals its long-term association with humans". Proceedings of the National Academy of Sciences of the United States of America. 115 (29): 7557–7562. Bibcode:2018PNAS..115.7557M. doi:10.1073/pnas.1804921115. PMC 6055166. PMID 29967156.
- ^ Pattison JR, Patou G (1996). Baron S, et al. (eds.). Parvoviruses. In: Barron's Medical Microbiology (4th ed.). Univ of Texas Medical Branch. ISBN 978-0-9631172-1-2.
- ^ Young NS, Brown KE (February 2004). "Parvovirus B19". The New England Journal of Medicine. 350 (6): 586–597. doi:10.1056/NEJMra030840. PMID 14762186. S2CID 33274873.
- ^ a b c "Parvovirus B19 and Pregnancy". CDC. Retrieved 25 February 2024.
- ^ a b c "Symptoms and Complications". CDC. Retrieved 25 February 2024.
- ^ "Fifth Disease (for Parents)".
- ^ a b Corcoran A, Doyle S (June 2004). "Advances in the biology, diagnosis and host-pathogen interactions of parvovirus B19" (PDF). Journal of Medical Microbiology. 53 (Pt 6): 459–475. doi:10.1099/jmm.0.05485-0. PMID 15150324.
- ^ Lehmann HW, von Landenberg P, Modrow S (June 2003). "Parvovirus B19 infection and autoimmune disease". Autoimmunity Reviews. 2 (4): 218–223. doi:10.1016/S1568-9972(03)00014-4. PMID 12848949.
- ^ Enders M, Weidner A, Enders G (May 2007). "Current epidemiological aspects of human parvovirus B19 infection during pregnancy and childhood in the western part of Germany". Epidemiology and Infection. 135 (4): 563–569. doi:10.1017/S095026880600731X. PMC 2870617. PMID 17064457.
- ^ a b c d Servey JT, Reamy BV, Hodge J (February 2007). "Clinical presentations of parvovirus B19 infection". American Family Physician. 75 (3): 373–376. PMID 17304869. Archived from the original on 21 August 2008. Retrieved 6 November 2009.
- ^ Ballou WR, Reed JL, Noble W, Young NS, Koenig S (February 2003). "Safety and immunogenicity of a recombinant parvovirus B19 vaccine formulated with MF59C.1". The Journal of Infectious Diseases. 187 (4): 675–678. doi:10.1086/368382. PMID 12599085.
- ^ a b Bernstein D, Sahly HE, Keitel W, Wolff M, Simone G, Segawa C, Wong S, Shelly D, Young N, Dempsey W (July 2011). "Safety and immunogenicity of a candidate parvovirus B19 vaccines". Vaccine. 29 (43): 7357–7363. doi:10.1016/j.vaccine.2011.07.080. PMC 3186820. PMID 21807052.
- ^ Lamont RF, Sobel JD, Vaisbuch E, Kusanovic JP, Mazaki-Tovi S, Kim SK, et al. (January 2011). "Parvovirus B19 infection in human pregnancy". BJOG. 118 (2): 175–186. doi:10.1111/j.1471-0528.2010.02749.x. PMC 3059196. PMID 21040396.
- ^ James, William D.; Berger, Timothy G.; et al. (2006). Andrews' Diseases of the Skin: clinical Dermatology. Saunders Elsevier. ISBN 978-0-7216-2921-6.
- ^ Aractingi, S.; Bakhos, D.; Flageul, B.; Vérola, O.; Brunet, M.; Dubertret, L.; Morinet, F. (October 1996). "Immunohistochemical and virological study of skin in the papular-purpuric gloves and socks syndrome". The British Journal of Dermatology. 135 (4): 599–602. doi:10.1111/j.1365-2133.1996.tb03839.x. ISSN 0007-0963. PMID 8915154.
- ^ Santonja C, Nieto-González G, Santos-Briz Á, Gutiérrez Zufiaurre M, Cerroni L, Kutzner H, Requena L (December 2011). "Immunohistochemical detection of parvovirus B19 in "gloves and socks" papular purpuric syndrome: direct evidence for viral endothelial involvement. Report of three cases and review of the literature". The American Journal of Dermatopathology. 33 (8): 790–795. doi:10.1097/DAD.0b013e318221bc41. PMID 22024574. S2CID 41097601.
- ^ Grilli, R.; Izquierdo, M.J.; Fariña, M.C.; Kutzner, H.; Gadea, I.; Martin, L.; Requena, L. (November 1999). "Papular-purpuric "gloves and socks" syndrome: Polymerase chain reaction demonstration of parvovirus B19 DNA in cutaneous lesions and sera". Journal of the American Academy of Dermatology. 41 (5): 793–796. doi:10.1016/S0190-9622(99)70027-7. PMID 10534650.
- ^ "Other Types of Arthritis". Arthritis Action UK. Arthritis Action. Archived from the original on 13 February 2016. Retrieved 16 October 2015.
- ^ Elling P, Olsson AT, Elling H (January 1996). "Synchronous variations of the incidence of temporal arteritis and polymyalgia rheumatica in different regions of Denmark; association with epidemics of Mycoplasma pneumoniae infection". The Journal of Rheumatology. 23 (1): 112–119. PMID 8838518.
- ^ Fjaerli HO, Vogt H, Bruu AL (September 1991). "[Human parvovirus B19 as the cause of aplastic crisis in hereditary spherocytosis]". Tidsskrift for den Norske Laegeforening. 111 (22): 2735–2737. PMID 1658972.
- ^ Beland SS, Daniel GK, Menard JC, Miller NM (September 1997). "Aplastic crisis associated with parvovirus B19 in an adult with hereditary spherocytosis". The Journal of the Arkansas Medical Society. 94 (4): 163–164. PMID 9308316.
- ^ Doldan Silvero AM, Acevedo-Gadea CR, Pantanowitz L, Dezube BJ, Johari V (June 2009). "Images in HIV/AIDS. Unsuspected parvovirus B19 infection in a person with AIDS". The AIDS Reader. 19 (6): 225–227. PMID 19642240.
- ^ Ergaz Z, Ornoy A (May 2006). "Parvovirus B19 in pregnancy". Reproductive Toxicology. 21 (4): 421–435. Bibcode:2006RepTx..21..421E. doi:10.1016/j.reprotox.2005.01.006. PMID 16580942.
- ^ Nagel HT, de Haan TR, Vandenbussche FP, Oepkes D, Walther FJ (January 2007). "Long-term outcome after fetal transfusion for hydrops associated with parvovirus B19 infection". Obstetrics and Gynecology. 109 (1): 42–47. doi:10.1097/01.AOG.0000249611.67873.94. PMID 17197586. S2CID 19731784.
- ^ Bassols AC (February 2008). "Parvovirus B19 and the new century". Clinical Infectious Diseases. 46 (4): 537–539. doi:10.1086/526523. PMID 18194096.
- ^ Katragadda L, Shahid Z, Restrepo A, Muzaffar J, Alapat D, Anaissie E (August 2013). "Preemptive intravenous immunoglobulin allows safe and timely administration of antineoplastic therapies in patients with multiple myeloma and parvovirus B19 disease". Transplant Infectious Disease. 15 (4): 354–360. doi:10.1111/tid.12067. PMID 23578205. S2CID 24400582.
- ^ a b Crabol Y, Terrier B, Rozenberg F, Pestre V, Legendre C, Hermine O, et al. (April 2013). "Intravenous immunoglobulin therapy for pure red cell aplasia related to human parvovirus b19 infection: a retrospective study of 10 patients and review of the literature". Clinical Infectious Diseases. 56 (7): 968–977. doi:10.1093/cid/cis1046. PMID 23243178.
- ^ Yang SH, Lin LW, Fang YJ, Cheng AL, Kuo SH (February 2012). "Parvovirus B19 infection-related acute hepatitis after rituximab-containing regimen for treatment of diffuse large B-cell lymphoma". Annals of Hematology. 91 (2): 291–294. doi:10.1007/s00277-011-1238-8. PMID 21538062. S2CID 6438572. Archived from the original on 14 March 2022. Retrieved 25 November 2021.
- ^ Klepfish A, Rachmilevitch E, Schattner A (November 2006). "Parvovirus B19 reactivation presenting as neutropenia after rituximab treatment". European Journal of Internal Medicine. 17 (7): 505–507. doi:10.1016/j.ejim.2006.05.002. PMID 17098597.
- ^ Hartmann JT, Meisinger I, Kröber SM, Weisel K, Klingel K, Kanz L (December 2006). "Progressive bicytopenia due to persistent parvovirus B19 infection after immunochemotherapy with fludarabine/cyclophosphamide and rituximab for relapsed B cell lymphoma". Haematologica. 91 (12 Suppl): ECR49. PMID 17194655.
- ^ Chandramouli S, Medina-Selby A, Coit D, Schaefer M, Spencer T, Brito LA, et al. (August 2013). "Generation of a parvovirus B19 vaccine candidate". Vaccine. 31 (37): 3872–3878. doi:10.1016/j.vaccine.2013.06.062. PMID 23827313.
