Aniline Yellow
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
4-(Phenyldiazenyl)aniline | |
| Other names
para-Aminoazobenzene
4-Phenylazoaniline AAB Brasilazina oil Yellow G Ceres Yellow Fast spirit Yellow Induline R Oil Yellow AAB Oil Yellow AN Oil Yellow B Oil Yellow 2G Oil Yellow R Organol Yellow Organol Yellow 2A Solvent Yellow Somalia Yellow 2G Stearix Brown 4R Sudan Yellow R Sudan Yellow RA C.I. 11000 | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.412 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
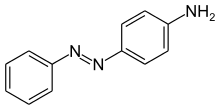
| C6H5N=NC6H4NH2 (C12H11N3) | |
| Molar mass | 197.24 g/mol |
| Density | 1.19 g/mL |
| Melting point | 123 to 126 °C (253 to 259 °F; 396 to 399 K) |
| Boiling point | > 360 °C (680 °F; 633 K) |
| Acidity (pKa) | 2.82 (25 ºC) |
| -118.3·10−6 cm3/mol | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Highly toxic Suspected carcinogen |
| GHS labelling: | |
 
| |
| Danger | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
200 mg/kg (mouse) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Aniline Yellow is a yellow azo dye and an aromatic amine. It is a derivative of azobenzene. It has the appearance of an orange powder. Aniline Yellow was the first azo dye. it was first produced in 1861 by C. Mene. The second azo dye was Bismarck Brown in 1863. Aniline Yellow was commercialized in 1864 as the first commercial azo dye, a year after aniline black. It is manufactured from aniline.
Uses
[edit]Aniline Yellow is used in microscopy for vital staining,[1] in pyrotechnics for yellow colored smokes, in yellow pigments and inks including inks for inkjet printers. It is also used in insecticides, lacquers, varnishes, waxes, oil stains, and styrene resins. It is also an intermediate in synthesis of other dyes, e.g. chrysoidine, indulines, Solid Yellow, and Acid Yellow.
Safety
[edit]Aminoazobenzene compounds are often carcinogenic.[2]
References
[edit]- ^ "Vital staining for protozoa".
- ^ Garg, Ashish; Bhat, Krishna L.; Bock, Charles W. (2002). "Mutagenicity of aminoazobenzene dyes and related structures: A QSAR/QPAR investigation". Dyes and Pigments. 55: 35–52. doi:10.1016/s0143-7208(02)00070-0.

