Dimanganese decacarbonyl

| |

| |
| Names | |
|---|---|
| IUPAC name
bis(pentacarbonylmanganese)(Mn—Mn)
| |
| Other names
Manganese carbonyl
Decacarbonyldimanganese | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.030.392 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Mn2(CO)10 | |
| Molar mass | 389.98 g/mol |
| Appearance | Yellow crystals |
| Density | 1.750 g/cm3 |
| Melting point | 154 °C (309 °F; 427 K) |
| Boiling point | sublimes 60 °C (140 °F; 333 K) at 0.5 mm Hg |
| Insoluble | |
| Structure[1] | |
| monoclinic | |
a = 14.14 Å, b = 7.10 Å, c = 14.63 Å α = 90°, β = 105.2°, γ = 90°
| |
Formula units (Z)
|
4 |
| 0 D | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
CO source |
| GHS labelling:[2] | |

| |
| Danger | |
| H301, H311, H331 | |
| P261, P264, P270, P271, P280, P301+P310, P302+P352, P304+P340, P311, P312, P321, P322, P330, P361, P363, P403+P233, P405, P501 | |
| Related compounds | |
Related compounds
|
Re2(CO)10 Co2(CO)8 Fe3(CO)12 Fe2(CO)9 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Dimanganese decacarbonyl,[3] which has the chemical formula Mn2(CO)10, is a binary bimetallic carbonyl complex centered around the first row transition metal manganese. The first reported synthesis of Mn2(CO)10 was in 1954 at Linde Air Products Company and was performed by Brimm, Lynch, and Sesny.[4] Their hypothesis about, and synthesis of, dimanganese decacarbonyl was fundamentally guided by the previously known dirhenium decacarbonyl (Re2(CO)10), the heavy atom analogue of Mn2(CO)10. Since its first synthesis, Mn2(CO)10 has been use sparingly as a reagent in the synthesis of other chemical species, but has found the most use as a simple system on which to study fundamental chemical and physical phenomena, most notably, the metal-metal bond. Dimanganese decacarbonyl is also used as a classic example to reinforce fundamental topics in organometallic chemistry like d-electron count, the 18-electron rule, oxidation state, valency,[5] and the isolobal analogy.
Synthesis
[edit]Many procedures have been reported for the synthesis of Mn2(CO)10 since 1954, the two most common general types are discussed herein. Some of these methods were not designed to create Mn2(CO)10, but rather treat Mn(I), Mn(II), or Mn(-I) as an oxidizing or reducing agent, respectively, for other species in the reaction, but produce Mn2(CO)10 nonetheless.
Reduction/carbonylation syntheses
[edit]The carbonylation route involves the reduction of a Mn(I) or Mn(II) salt to the Mn(0) species in concert with carbonylation to a coordinatively saturated metal center with CO gas. The carbonylation using CO can be under heightened pressures of CO, relative to atmospheric pressure, or at ambient pressure. Examples of each are given.
High pressure carbonylation
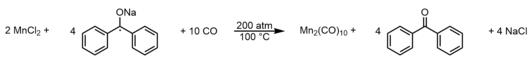
[edit]As previously mentioned, Mn2(CO)10 was first prepared in 1954 by Brimm, Lynch, and Sesny, albeit in yields of ~1%, by the reduction of manganese(II) iodide with magnesium(0) under 3000 psi (~200 atm) of carbon monoxide (CO).[4] The balanced reaction is represented by:A more efficient preparation was developed in 1958 and entails reduction of anhydrous manganese(II) chloride with sodium benzophenone ketyl radical under similarly high pressures (200 atm) of CO.[6] This method yielded ~32% of the dimanganese decacarbonyl complex, producing enough material for the first real opportunities to rigorously study the chemical and physical properties of the molecule. This method is represented by the balanced equation:
Low pressure carbonylation
[edit]Despite successes in the synthesis of Mn2(CO)10, the safety concerns and limited batch size surrounding high pressure carbonylation methods necessitated alternative, low pressure procedures to obtain the target compound. In 1968, the first ambient CO pressure carbonylation synthesis of Mn2(CO)10 was reported from the commercially available and inexpensive methylcyclopentadienyl manganese tricarbonyl (MMT) and sodium(0) as the reductant.[7] The balanced equation being:The efficiency of the method ranged from 16 to 20% yield, lower than what was previously reported, however, it could be performed more safely and on mole scale.
Dimerization syntheses
[edit]The second overarching method used to make Mn2(CO)10 is similar to the first in that it usually requires alteration of a Mn(I), or in this case, Mn(-I) to the corresponding Mn(0) species. These preparations differ, however, by beginning with manganese precursors, sometimes commercially available, that need no additional CO ligands and simply dimerize to form the target molecule. This poses the significant logistic and safety advantage of not dealing with toxic CO gas and is the prevailing general method for the academic synthesis of Mn2(CO)10.
The first explicit success in this area was published in 1977, which featured a pentacarbonylhydridomanganese(I) Mn source, with Se(PF2)2 as the reductant.[8] The balanced equation for this transformation is:Alterations of the terminal reductant have been reported in the manganese hydride case.[9][10][11] Similar methods exist for Mn(CO)5X compounds where X = Cl, Br, or I, and more rarely, Mn(CO)6 bound with a weakly coordinating anion.[12][13][14][15][16][17] Using similar logic, stable salts of the pentacarbonyl manganate anion can also be employed with an oxidant to access the same Mn2(CO)10 complex.[18][19][20] An example of this is the reduction of triphenylcyclopropenium tetrafluoroborate with sodium pentacarbonyl manganate to produce the dimer of each.[21] The balanced equation is given by:

One additional interesting synthesis of Mn2(CO)10 occurs by combination of a hexacarbonylmanganese(I) tetrafluoroborate salt with a sodium pentacarbonyl manganate salt. In this instance, manganese is both the oxidant and reductant, producing two formal Mn(0) atoms.[22] The balanced equation is:
Structure and bonding
[edit]High precision crystallographic and theoretical studies of the physical and electronic structures of Mn2(CO)10 have been performed and are discussed with respect to the published literature below, however, a qualitative approach can also be taken to predict its constitutional structure using fundamental principles of inorganic and organometallic chemistry.
The stoichiometric composition of Mn2(CO)10, derived from elemental analysis, informs a 5:1 ratio of CO to Mn. The assumed binary carbonyl complex given this information is pentacarbonylmanganese(0). However, the sum of the d-electron count (7 for Mn(0)) and the electron contributions from the ligands (10 for 5 CO) yields a 17-electron, metalloradical complex for Mn(CO)5. This is a highly unstable configuration, isolobal to the methyl radical, which can be expected to homodimerize to the constitutionally symmetric dinuclear complex in order for both Mn nuclei to achieve an 18-electron, noble gas configuration. Indeed, the true structure of the Mn(0) binary carbonyl structure is a dimeric, dinuclear complex.
Crystal structure
[edit]This hypothesized structure was confirmed explicitly through x-ray diffraction studies, first in two dimensions in 1957,[23] followed by its single crystal three-dimensional analysis in 1963.[24] The crystal structure of Mn2(CO)10 was redetermined at high precision at room temperature in 1981 and bond lengths mentioned herein refer to results from that study.[25] Mn2(CO)10 has no bridging CO ligands: it can be described as containing two axially-linked (CO)5Mn- subunits. These Mn subunits are spaced at a distance of 290.38(6) pm, a bonding distance that is longer than that predicted by Pekka Pyykko.[26] There are two kinds of CO ligands; one CO linked to each Mn atom that is coaxial with the Mn-Mn bond and four “equatorial” carbonyls bonded to each Mn atom that are nearly perpendicular to the Mn-Mn bond (Mn’-Mn-CO(equatorial) angles range from 84.61(7) to 89.16(7) degrees). The axial carbonyl distance of (181.1 pm) is 4.5 pm shorter than the average equatorial manganese-carbonyl distance of 185.6 pm. In the stable rotamer, the two Mn(CO)5 subunits are staggered. Thus, the overall molecule has approximate point group D4d symmetry, which is an uncommon symmetry shared with S2F10. The Mn2(CO)10 molecule is isomorphous with the other group 7 binary metal carbonyls Tc2(CO)10 and Re2(CO)10.

Electronic structure
[edit]Initial fundamental experimental and theoretical studies on the electronic structure of Mn2(CO)10 were performed used a mixture of photoelectron spectroscopy, infrared spectroscopy, and an iterative extended-Hückel-type molecular orbital calculation.[27][28] The electronic structure of Mn2(CO)10 was most reported in 2017 using the BP86D functional with TZP basis set.[29] The electronic structure described herein, along with relevant orbital plots, are reproduced from the methods used in that study using Orca (5.0.3)[30] and visualized using IBOView (v20150427).[31] The two main interactions of interest in the system are the metal-to-ligand pi-backbonding interactions and the metal-metal sigma bonding orbital. The pi-backbonding interactions illustrated below occur between the t2g d-orbital set and the CO π* antibonding orbitals. The degenerate dxz and dyz backbonding interactions with both axial and equatorial CO ligands is the HOMO-15. More total delocalization occurs onto the axial CO antibonding orbital than does the equatorial, which is thought to rationalize the shorter Mn-C bond length.
- Two degenerate pi-backbonding orbitals of dimanganese decacarbonyl
The primary Mn-Mn σ-bonding orbital is composed of two dz2 orbitals, represented by the HOMO-9.

Other large contributions made in this area were by Ahmed Zewail using ultrafast, femtosecond spectroscopy en route to his 1999 Nobel Prize.[32] His discoveries elucidated much about the time scales and energies associated with the molecular motions of Mn2(CO)10, as well as the Mn-Mn and Mn-C bond cleavage events.[33]
Reactivity
[edit]Mn2(CO)10 is air stable as a crystalline solid, but solutions require Schlenk techniques. Mn2(CO)10 is chemically active at both the Mn-Mn and Mn-CO bonds due to low, and surprisingly similar, bond dissociation energies of ~36 kcal/mol (151 kJ/mol)[34] and ~38 kcal/mol (160 kJ/mol),[35] respectively. For this reason, reactivity can happen at either site of the molecule, sometimes selectively. Examples of each are given.
Mn-Mn bond cleavage reactions
[edit]The Mn-Mn bond is sensitive to both oxidation and reduction, producing two equivalents of the corresponding Mn(I) and Mn(-I) species, respectively. Both of the potential resultant species can be derived further. Redox neutral cleavage is possible both thermally and photochemically, producing two equivalents of the Mn(0) radical. Examples of each are given below.
Oxidative cleavage
[edit]Selective mono-oxidation of the Mn-Mn bond is most often done via addition of classical metal oxidants (e.g. CeIV, PbIV, etc) or weak homonuclear single covalent bonds of the form X-X (X is group 16 or 17 element).[36][37][38][39][40] These reactions yield the [Mn(CO)5]+ cation with a bound weakly coordinating anion, or the Mn(CO)5X complex. The general reaction schemes for each are seen as balanced equations below:or for two-electron oxidantsandfor E = O, S, Se, Tefor X = F, Cl, Br, I
Reductive cleavage
[edit]Reductive cleavage is almost always done with sodium metal,[41][42] yielding the [Mn(CO)5]− anion with the sodium counterion. The balanced general reactions are given below:The resultant manganate anion is a potent nucleophile, which can be protonated to give the manganese hydride,[43][44] or alkylated with organic halides[45][46][43] to give a large swath of organomanganese(I) complexes.
Redox-neutral cleavage
[edit]Homolytic cleavage, usually via light,[47] but sometimes heat,[48] gives the Mn(0) metalloradical, which can react with itself to reform Mn2(CO)10, or combine with other radical species that usually result in formal oxidation to Mn(I). This reactivity is comparable to that of organic, carbon-based radicals via the isolobal analogy. The homolytic cleavage is given by:The use of the produced radical species, [Mn(CO)5]*, has found several applications as a radical initiator for various organic methodologies[49][50][51] and polymerization reactions.[52][53][54]
Ligand substitution reactions
[edit]Ligand substitution reactions that do not disrupt the Mn-Mn bonding is done by using strongly sigma donating L-type ligands that can outcompete CO without participating in redox reactivity.[55] This requirement usually necessitates phosphines[56][57] or N-heterocyclic carbenes (NHCs),[58] with substitution occurring at the axial position according to the reactions below:

Safety
[edit]Mn2(CO)10 is a volatile source of a metal and a source of CO.
References
[edit]- ^ Melvyn Rowen Churchill, Kwame N. Amoh, and Harvey J. Wasserman (1981). "Redetermination of the crystal structure of dimanganese decacarbonyl and determination of the crystal structure of dirhenium decacarbonyl. Revised values for the manganese-manganese and rhenium-rhenium bond lengths in dimanganese decacarbonyl and dirhenium decacarbonyl". Inorg. Chem. 20 (5): 1609–1611. doi:10.1021/ic50219a056.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "Decacarbonyldimanganese". pubchem.ncbi.nlm.nih.gov. Retrieved 27 December 2021.
- ^ Pauson, Peter L.; Friestad, Gregory K. (2009-09-15), "Decacarbonyldimanganese", in John Wiley & Sons, Ltd (ed.), Encyclopedia of Reagents for Organic Synthesis, Chichester, UK: John Wiley & Sons, Ltd, pp. rd001.pub2, doi:10.1002/047084289x.rd001.pub2, ISBN 978-0-471-93623-7, retrieved 2023-03-12
- ^ a b Brimm, E. O.; Lynch, M. A.; Sesny, W. J. (July 1954). "Preparation and Properties of Manganese Carbonyl". Journal of the American Chemical Society. 76 (14): 3831–3835. doi:10.1021/ja01643a071. ISSN 0002-7863.
- ^ Parkin, Gerard (May 2006). "Valence, Oxidation Number, and Formal Charge: Three Related but Fundamentally Different Concepts". Journal of Chemical Education. 83 (5): 791. Bibcode:2006JChEd..83..791P. doi:10.1021/ed083p791. ISSN 0021-9584.
- ^ Closson, Rex D.; Buzbee, Lloyd R.; Ecke, George G. (December 1958). "A New Metal Carbonyl Synthesis 1". Journal of the American Chemical Society. 80 (23): 6167–6170. doi:10.1021/ja01556a005. ISSN 0002-7863.
- ^ King, R. B.; Stokes, J. C.; Korenowski, T. F. (1968-01-01). "A convenient synthesis of dimanganese decarbonyl from inexpensive starting materials at atmospheric pressure". Journal of Organometallic Chemistry. 11: 641–643. doi:10.1016/0022-328X(68)80099-3. ISSN 0022-328X.
- ^ Arnold, David E. J.; Cromie, Ernest R.; Rankin, David W. H. (1977). "Preparation and chemical and spectroscopic properties of bis(difluorophosphino) selenide". Journal of the Chemical Society, Dalton Transactions (20): 1999–2004. doi:10.1039/dt9770001999. ISSN 0300-9246.
- ^ Sweany, Ray; Butler, Steven C.; Halpern, Jack (1981-06-23). "The hydrogenation of 9,10-dimethylanthracene by hydridopentacarbonylmanganese(I). Evidence for a free-radical mechanism". Journal of Organometallic Chemistry. 213 (2): 487–492. doi:10.1016/S0022-328X(00)82954-X. ISSN 0022-328X.
- ^ Booth, Brian L.; Haszeldine, Robert N.; Holmes, Robert G. G. (1982). "Reactions involving transition metals. Part 16. Rhodium, iridium, platinum, and gold complexes containing the bis(trifluoromethyl)amino-oxy-ligand". Journal of the Chemical Society, Dalton Transactions (3): 523–529. doi:10.1039/dt9820000523. ISSN 0300-9246.
- ^ Tam, Wilson; Marsi, Marianne; Gladysz, J. A. (May 1983). "Bimetallic anionic formyl complexes: synthesis and properties". Inorganic Chemistry. 22 (10): 1413–1421. doi:10.1021/ic00152a001. ISSN 0020-1669.
- ^ Kolthammer, Brian W. S.; Legzdins, Peter (1978). "Organometallic nitrosyl chemistry. Part 3. Some aspects of the chemistry of bis[(η-cyclopentadienyl)dinitrosylchromium]". J. Chem. Soc., Dalton Trans. (1): 31–35. doi:10.1039/DT9780000031. ISSN 0300-9246.
- ^ Manning, Mark C.; Trogler, William C. (1981-01-01). "Reduction of metal carbonyls with alkali metal carbides". Inorganica Chimica Acta. 50: 247–250. doi:10.1016/S0020-1693(00)83752-4. ISSN 0020-1693.
- ^ Gibson, Dorothy H.; Hsu, Wen-Liang (1982-01-01). "Reactions of manganese carbonyls with quaternary ammonium halides". Inorganica Chimica Acta. 59: 93–99. doi:10.1016/S0020-1693(00)87314-4. ISSN 0020-1693.
- ^ Kuchynka, D. J.; Amatore, C.; Kochi, J. K. (November 1986). "Manganese(0) radicals and the reduction of cationic carbonyl complexes: selectivity in the ligand dissociation from 19-electron species". Inorganic Chemistry. 25 (23): 4087–4097. doi:10.1021/ic00243a009. ISSN 0020-1669.
- ^ Geier, Jens; Willner, Helge; Lehmann, Christian W.; Aubke, Friedhelm (2007-08-01). "Formation of Hexacarbonylmanganese(I) Salts, [Mn(CO) 6 ] + X - , in Anhydrous HF". Inorganic Chemistry. 46 (17): 7210–7214. doi:10.1021/ic700798z. ISSN 0020-1669. PMID 17616186.
- ^ Manning, Peter J.; Peterson, Louis K.; Wada, Fumio; Dhami, Randi S. (1986-04-01). "Synthesis and reactions of [M(CO)4(Ph2PSiMe3)X] complexes (M = Mn, Re; X = Halogen)". Inorganica Chimica Acta. 114 (1): 15–20. doi:10.1016/S0020-1693(00)84581-8. ISSN 0020-1693.
- ^ DuBois, Donn A.; Duesler, Eileen N.; Paine, Robert T. (January 1985). "Formation and x-ray crystal structure determination of an unusual phosphorus-phosphorus coupled bicyclodiphosphazane complex". Inorganic Chemistry. 24 (1): 3–5. doi:10.1021/ic00195a003. ISSN 0020-1669.
- ^ Ozin, Geoffrey A.; Coleson, Kraig M.; Huber, Helmut X. (March 1983). "Reactions of solvated metal atoms with organometallic complexes in solution. A metal atom microsolution spectroscopic and synthetic study of the reaction pathways available to singly metal-metal bonded organometallic complexes and their organometallic anions". Organometallics. 2 (3): 415–420. doi:10.1021/om00075a011. ISSN 0276-7333.
- ^ Müller, Manfred; Vahrenkamp, Heinrich (June 1983). "Cluster‐Konstruktion: Schrittweiser Aufbau von μ 3 ‐RP‐Trimetall‐Clustern über P–Hal‐Verbindungen". Chemische Berichte. 116 (6): 2322–2336. doi:10.1002/cber.19831160621. ISSN 0009-2940.
- ^ Hughes, Russell P.; Lambert, James M. J.; Reisch, John W.; Smith, Wayne L. (October 1982). "Reinvestigations of some reactions of metal carbonyl anions with cyclopropenium cations. Conversion of .eta.3-cyclopropenyl to .eta.3-cyclobutenonyl ligands". Organometallics. 1 (10): 1403–1405. doi:10.1021/om00070a027. ISSN 0276-7333.
- ^ Lee, K. Y.; Kuchynka, D. J.; Kochi, J. K. (1987-09-01). "Formation of metal-metal bonds by ion-pair annihilation. Dimanganese carbonyls from manganate(I-) anions and manganese(I) cations". Organometallics. 6 (9): 1886–1897. doi:10.1021/om00152a010. ISSN 0276-7333.
- ^ Dahl, Lawrence F.; Ishishi, Etsuro; Rundle, R. E. (2004-10-06). "Polynuclear Metal Carbonyls. I. Structures of Mn2(CO)10 and Re2(CO)10". The Journal of Chemical Physics. 26 (6): 1750. doi:10.1063/1.1743615. ISSN 0021-9606.
- ^ Dahl, L. F.; Rundle, R. E. (1963-05-10). "The crystal structure of dimanganese decacarbonyl Mn2(CO)10". Acta Crystallographica. 16 (5): 419–426. doi:10.1107/S0365110X63001080. ISSN 0365-110X.
- ^ Churchill, Melvyn Rowen; Amoh, Kwame N.; Wasserman, Harvey J. (May 1981). "Redetermination of the crystal structure of dimanganese decacarbonyl and determination of the crystal structure of dirhenium decacarbonyl. Revised values for the manganese-manganese and rhenium-rhenium bond lengths in dimanganese decacarbonyl and dirhenium decacarbonyl". Inorganic Chemistry. 20 (5): 1609–1611. doi:10.1021/ic50219a056. ISSN 0020-1669.
- ^ Pyykkö, Pekka (2015-03-19). "Additive Covalent Radii for Single-, Double-, and Triple-Bonded Molecules and Tetrahedrally Bonded Crystals: A Summary". The Journal of Physical Chemistry A. 119 (11): 2326–2337. Bibcode:2015JPCA..119.2326P. doi:10.1021/jp5065819. ISSN 1089-5639. PMID 25162610.
- ^ Levenson, Robert A.; Gray, Harry B.; Ceasar, Gerald P. (June 1970). "Electronic and vibrational spectroscopy in a nematic liquid crystal solvent. Band polarizations of binuclear metal carbonyls". Journal of the American Chemical Society. 92 (12): 3653–3658. doi:10.1021/ja00715a018. ISSN 0002-7863.
- ^ Levenson, Robert A.; Gray, Harry B. (October 1975). "Electronic structure of compounds containing metal-metal bonds. Decacarbonyldimetal and related complexes". Journal of the American Chemical Society. 97 (21): 6042–6047. doi:10.1021/ja00854a015. ISSN 0002-7863.
- ^ Menacer, Rafik; May, Abdelghani; Belkhiri, Lotfi; Mousser, Abdelhamid (2017-11-28). "Electronic structure and bonding of the dinuclear metal M2(CO)10 decacarbonyls: applications of natural orbitals for chemical valence". Journal of Molecular Modeling. 23 (12): 358. doi:10.1007/s00894-017-3523-5. ISSN 0948-5023. PMID 29185066. S2CID 3814626.
- ^ Neese, Frank (January 2012). "The ORCA program system". WIREs Computational Molecular Science. 2 (1): 73–78. doi:10.1002/wcms.81. ISSN 1759-0876. S2CID 62137389.
- ^ Knizia, Gerald (2013-11-12). "Intrinsic Atomic Orbitals: An Unbiased Bridge between Quantum Theory and Chemical Concepts". Journal of Chemical Theory and Computation. 9 (11): 4834–4843. arXiv:1306.6884. doi:10.1021/ct400687b. ISSN 1549-9618. PMID 26583402. S2CID 17717923.
- ^ Zewail, Ahmed H. (2001), "Femtochemistry: Atomic-Scale Dynamics of the Chemical Bond Using Ultrafast Lasers (Nobel Lecture)", Femtochemistry, Weinheim, FRG: Wiley-VCH Verlag GmbH, pp. 1–85, doi:10.1002/3527600183.ch1, ISBN 352730259X, retrieved 2023-03-13
- ^ Kyu Kim, Sang; Pedersen, Soren; Zewail, Ahmed H. (1995-02-24). "Femtochemistry of organometallics: dynamics of metal-metal and metal-ligand bond cleavage in M2(CO)10". Chemical Physics Letters. 233 (5): 500–508. Bibcode:1995CPL...233..500K. doi:10.1016/0009-2614(95)00050-E. ISSN 0009-2614.
- ^ Hughey, Joseph L.; Anderson, Craig P.; Meyer, Thomas J. (1977-02-01). "Photochemistry of Mn2(CO)10". Journal of Organometallic Chemistry. 125 (2): C49 – C52. doi:10.1016/S0022-328X(00)89455-3. ISSN 0022-328X.
- ^ Smith, G. P. (1988-01-01). "Gas-phase first bond dissociation energies in transition-metal carbonyls". Polyhedron. 7 (16): 1605–1608. doi:10.1016/S0277-5387(00)81785-4. ISSN 0277-5387.
- ^ Davidson, J. L.; Sharp, D. W. A. (1973-01-01). "Metal perfluoro-alkyl- and -aryl-thiolates. Part II. Molybdenum, tungsten, manganese, iron, and nickel derivatives". Journal of the Chemical Society, Dalton Transactions (19): 1957–1960. doi:10.1039/DT9730001957. ISSN 1364-5447.
- ^ Chaudhuri, M. K.; Haas, A.; Wensky, A. (1976-08-24). "Photoinduced reactions of (CF3S)3N and CF3SeSeCF3 with Mn2(CO)10 and Fe(CO)5". Journal of Organometallic Chemistry. 116 (3): 323–326. doi:10.1016/S0022-328X(00)94469-3. ISSN 0022-328X.
- ^ Arsenault, Clément; Bougeard, Peter; Sayer, Brian G.; Yeroushalmi, Shahin; McGlinchey, Michael J. (1984-04-17). "[η5-1,2,3,4,5-pentakis(carbomethoxy)cyclopentadienyl]tricarbonylmanganese(I): Synthesis, spectroscopy and reactivity". Journal of Organometallic Chemistry. 265 (3): 283–290. doi:10.1016/0022-328X(84)80092-3. ISSN 0022-328X.
- ^ Schmidt, Steven P.; Trogler, William C.; Basolo, Fred (March 1984). "Mechanism of halogenation of dimanganese, manganese-rhenium, and dirhenium decacarbonyls". Journal of the American Chemical Society. 106 (5): 1308–1313. doi:10.1021/ja00317a023. ISSN 0002-7863.
- ^ Hernández, José G.; Butler, Ian S.; Friščić, Tomislav (2014-06-16). "Multi-step and multi-component organometallic synthesis in one pot using orthogonal mechanochemical reactions". Chemical Science. 5 (9): 3576. doi:10.1039/C4SC01252F. ISSN 2041-6520.
- ^ Warnock, Garry F. P.; Moodie, Lyn Cammarano; Ellis, John E. (March 1989). "Highly reduced organometallics. Part 25. Reactions of trisodium tetracarbonylmetalates(3-) of manganese and rhenium with Broensted acids and other electrophiles. Synthesis of H2M(CO)4- (M = Mn and Re), (CH3)2Re(CO)4-, the first dialkyl derivative of a carbonylmetalate trianion, and related anionic species". Journal of the American Chemical Society. 111 (6): 2131–2141. doi:10.1021/ja00188a029. ISSN 0002-7863.
- ^ Kuchynka, D. J.; Kochi, J. K. (March 1989). "Equilibrium of 17-electron and 19-electron organometallic radicals derived from carbonylmanganese anions and cations". Inorganic Chemistry. 28 (5): 855–863. doi:10.1021/ic00304a012. ISSN 0020-1669.
- ^ a b Nappa, Mario J.; Santi, Roberto; Halpern, Jack (January 1985). "Mechanisms of the carbon-hydrogen bond-forming binuclear reductive elimination reactions of benzyl- and hydridomanganese carbonyls". Organometallics. 4 (1): 34–41. doi:10.1021/om00120a007. ISSN 0276-7333.
- ^ Wassink, Berend; Thomas, Marian J.; Wright, Steven C.; Gillis, Daniel J.; Baird, Michael C. (April 1987). "Mechanisms of the hydrometalation (insertion) and stoichiometric hydrogenation reactions of conjugated dienes effected by manganese pentacarbonyl hydride: processes involving the radical pair mechanism". Journal of the American Chemical Society. 109 (7): 1995–2002. doi:10.1021/ja00241a016. ISSN 0002-7863.
- ^ Casey, Charles P.; Scheck, Daniel M. (April 1980). "Mechanism of reductive elimination of acetophenone from Me4+[cis-(CO)4Mn(COMe)(COPh)]-". Journal of the American Chemical Society. 102 (8): 2728–2731. doi:10.1021/ja00528a034. ISSN 0002-7863.
- ^ Benson, Ian B.; Hunt, James; Knox, Selby A. R.; Oliphant, Valerie (1978). "Organosulphur–transition-metal chemistry. Part 1. Reactions of carbon disulphide with metal carbonyl anions". J. Chem. Soc., Dalton Trans. (10): 1240–1246. doi:10.1039/DT9780001240. ISSN 0300-9246.
- ^ Herrick, Richard S.; Brown, Theodore L. (December 1984). "Flash photolytic investigation of photoinduced carbon monoxide dissociation from dinuclear manganese carbonyl compounds". Inorganic Chemistry. 23 (26): 4550–4553. doi:10.1021/ic00194a028. ISSN 0020-1669.
- ^ Wegman, R. W.; Olsen, R. J.; Gard, D. R.; Faulkner, L. R.; Brown, Theodore L. (October 1981). "Flash photolysis study of the metal-metal bond homolysis in dimanganese decacarbonyl and dirhenium decacarbonyl". Journal of the American Chemical Society. 103 (20): 6089–6092. doi:10.1021/ja00410a017. ISSN 0002-7863.
- ^ Gilbert, Bruce C.; Kalz, Wilhelm; Lindsay, Chris I.; McGrail, P. Terry; Parsons, Andrew F.; Whittaker, David T. E. (1999-08-13). "Radical cyclisations promoted by dimanganese decacarbonyl: A new and flexible approach to 5-membered N-heterocycles". Tetrahedron Letters. 40 (33): 6095–6098. doi:10.1016/S0040-4039(99)01271-X. ISSN 0040-4039.
- ^ Gilbert, Bruce C.; Parsons, Andrew F. (2002-02-25). "The use of free radical initiators bearing metal–metal, metal–hydrogen and non-metal–hydrogen bonds in synthesis". Journal of the Chemical Society, Perkin Transactions 2 (3): 367–387. doi:10.1039/B102044G. ISSN 1364-5471.
- ^ Fukuyama, Takahide; Nishitani, Satoshi; Inouye, Takaya; Morimoto, Keisuke; Ryu, Ilhyong (2006-03-01). "Effective Acceleration of Atom Transfer Carbonylation of Alkyl Iodides by Metal Complexes. Application to the Synthesis of the Hinokinin Precursor and Dihydrocapsaicin". Organic Letters. 8 (7): 1383–1386. doi:10.1021/ol060123+. ISSN 1523-7060. PMID 16562897.
- ^ Ciftci, Mustafa; Tasdelen, Mehmet Atilla; Yagci, Yusuf (2013-12-11). "Sunlight induced atom transfer radical polymerization by using dimanganese decacarbonyl". Polymer Chemistry. 5 (2): 600–606. doi:10.1039/C3PY01009K. ISSN 1759-9962.
- ^ Gilbert, Bruce C.; Harrison, Richard J.; Lindsay, Chris I.; McGrail, P. Terry; Parsons, Andrew F.; Southward, Richard; Irvine, Derek J. (2003-12-01). "Polymerization of Methyl Methacrylate Using Dimanganese Decacarbonyl in the Presence of Organohalides". Macromolecules. 36 (24): 9020–9023. Bibcode:2003MaMol..36.9020G. doi:10.1021/ma034712w. ISSN 0024-9297.
- ^ Ciftci, Mustafa; Norsic, Sébastien; Boisson, Christophe; D'Agosto, Franck; Yagci, Yusuf (May 2015). "Synthesis of Block Copolymers Based on Polyethylene by Thermally Induced Controlled Radical Polymerization Using Mn 2 (CO) 10". Macromolecular Chemistry and Physics. 216 (9): 958–963. doi:10.1002/macp.201500016.
- ^ Coville, N. J.; Stolzenberg, A. M.; Muetterties, E. L. (April 1983). "Mechanism of ligand substitution in dimanganese decacarbonyl". Journal of the American Chemical Society. 105 (8): 2499–2500. doi:10.1021/ja00346a079. ISSN 0002-7863.
- ^ Herrinton, Thomas; Brown, Theodore (October 1, 1985). "Substitution of manganese pentacarbonyl is associative". Journal of the American Chemical Society. 107 (20): 5700–5703. doi:10.1021/ja00306a016.
- ^ Reimann, Rolf H.; Singleton, Eric (1976-01-01). "Reactions of metal carbonyls. Part 7. Substitution reactions of decacarbonyldimanganese with tertiary phosphorus and arsenic ligands". Journal of the Chemical Society, Dalton Transactions (20): 2109–2114. doi:10.1039/DT9760002109. ISSN 1364-5447.
- ^ Fraser, Roan; van Sittert, Cornelia G. C. E.; van Rooyen, Petrus H.; Landman, Marilé (2017-05-01). "Synthesis and structural investigation of mono- and dimetallic N-heterocyclic carbene complexes of group VII transition metals". Journal of Organometallic Chemistry. 835: 60–69. doi:10.1016/j.jorganchem.2017.02.031. ISSN 0022-328X.


![{\displaystyle {\ce {2 Mn(\eta^5-(CH3)C5H4)(CO)3 + 2 Na + 4 CO -> Mn2(CO)10 + 2Na[(CH3)C5H4]}}}](https://wikimedia.riteme.site/api/rest_v1/media/math/render/svg/7e0469b5df58316feaed1f33805bb4c7d4e6f4c2)

 + Na[Mn(CO)5] -> Mn2(CO)10 + Na[BF4] + CO}}}](https://wikimedia.riteme.site/api/rest_v1/media/math/render/svg/a0bc208f17e2a730d87ae834a00dae8270897344)






![{\displaystyle {\ce {Mn2(CO)10 + 2 Na^{0}-> 2Na[Mn(CO)5]}}}](https://wikimedia.riteme.site/api/rest_v1/media/math/render/svg/47f1b62210d70cf7a7f82ee3b1d7791a8314dd14)
![{\displaystyle {\ce {Mn2(CO)10 + h\nu -> 2[Mn(CO)5]^{.}}}}](https://wikimedia.riteme.site/api/rest_v1/media/math/render/svg/649cb57e761488981ed55970fb04937016b7288b)