Citrulline
| |

| |
| Names | |
|---|---|
| IUPAC name
2-Amino-5-(carbamoylamino)pentanoic acid[1]
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| 1725417, 1725415 D, 1725416 L | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.006.145 |
| EC Number |
|
| 774677 L | |
| KEGG | |
| MeSH | Citrulline |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H13N3O3 | |
| Molar mass | 175.188 g·mol−1 |
| Appearance | White crystals |
| Odor | Odourless |
| log P | −1.373 |
| Acidity (pKa) | 2.508 |
| Basicity (pKb) | 11.489 |
| Thermochemistry | |
Heat capacity (C)
|
232.80 J K−1 mol−1 |
Std molar
entropy (S⦵298) |
254.4 J K−1 mol−1 |
| Related compounds | |
Related alkanoic acids
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
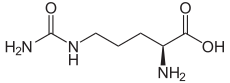
The organic compound citrulline is an α-amino acid.[2] Its name is derived from citrullus, the Latin word for watermelon. Although named and described by gastroenterologists since the late 19th century, it was first isolated from watermelon in 1914 by Japanese researchers Yatarō Koga (古賀彌太郎) and Ryō Ōtake (大嶽了)[3][4] and further codified by Mitsunori Wada of Tokyo Imperial University in 1930.[5][6][7] It has the formula H2NC(O)NH(CH2)3CH(NH2)CO2H. It is a key intermediate in the urea cycle, the pathway by which mammals excrete ammonia by converting it into urea. Citrulline is also produced as a byproduct of the enzymatic production of nitric oxide from the amino acid arginine, catalyzed by nitric oxide synthase.[8]
Biosynthesis
[edit]Citrulline can be derived from:
- from arginine via nitric oxide synthase, as a byproduct of the production of nitric oxide for signaling purposes
- from ornithine through the breakdown of proline or glutamine/glutamate
- from asymmetric dimethylarginine via DDAH
Citrulline is made from ornithine and carbamoyl phosphate in one of the central reactions in the urea cycle. It is also produced from arginine as a byproduct of the reaction catalyzed by NOS family (NOS; EC 1.14.13.39).[9] It is also prevalent in trichohyalin at the inner root sheath and medulla of hair follicles, where it is synthesized from arginine.[10] Arginine is first oxidized into N-hydroxyl-arginine, which is then further oxidized to citrulline concomitant with release of nitric oxide.
Citrulline is also made by enterocytes of the small intestine.[2][11]
Function
[edit]Citrulline is a metabolic intermediate within the urea cycle, which is the pathway by which mammals excrete ammonia by converting it into urea. Citrulline is also produced as a byproduct of the enzymatic production of nitric oxide from the amino acid arginine, catalyzed by nitric oxide synthase. In the yeast species Saccharomyces cerevisiae, citrulline is a metabolic intermediate in the latter, cytosolic half of the arginine biosynthesis pathway.[12]
Several proteins contain citrulline as a result of a post-translational modification. These citrulline residues are generated by a family of enzymes called peptidylarginine deiminases (PADs), which convert arginine into citrulline in a process called citrullination or deimination with the help of calcium ions. Proteins that normally contain citrulline residues include myelin basic protein (MBP), filaggrin, and several histone proteins, whereas other proteins, such as fibrin and vimentin are susceptible to citrullination during cell death and tissue inflammation.
Circulating citrulline concentration is a biomarker of intestinal functionality.[13][14]
Commercial use
[edit]L-citrulline is sold as a dietary supplement, usually in powder form. Advocates suggest that since L-citrulline boosts nitric oxide production, helping arterial function and thus blood flow throughout the body, this might be helpful in treating or preventing various ailments.
- Lowering blood pressure in people with elevated blood pressure.
- Easing symptoms of mild-to-moderate erectile dysfunction, with fewer risks than Viagra.
- Ameliorating circulatory problems such as slow wound healing due to diabetes.
- Raising muscle protein levels, preventing malnourishment in the elderly and possibly improving athletic performance.
- Treating intestinal problems, including short bowel syndrome, celiac disease and radiation-caused small bowel damage
- Various genetic disorders and health conditions such as liver disease, Parkinson's disease and certain dementias.
These assertions are based on animal studies, and like most dietary supplement claims have not be endorsed by the Food and Drugs Administration.[15]
L-citrulline is added to certain energy drinks such as Reign and touted for its thermogenic effects.[16]
See also
[edit]References
[edit]- ^ "Citrulline - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 16 September 2004. Identification. Retrieved 1 May 2012.
- ^ a b Banerjee, Aryamitra (2014-01-01), Gupta, Ramesh C. (ed.), "Chapter 15 - Gastrointestinal toxicity biomarkers", Biomarkers in Toxicology, Boston: Academic Press, pp. 269–277, doi:10.1016/b978-0-12-404630-6.00015-4, ISBN 978-0-12-404630-6, S2CID 88798984, retrieved 2020-11-10
- ^ Koga, Yatarō; Ōtake, Ryō (1914). "Study report on the constituents of squeezed watermelon". Tokyo Kagaku Kaishi [Journal of the Tokyo Chemical Society]. 35 (5): 519–528. doi:10.1246/nikkashi1880.35.519.
- ^ Fragkos, Konstantinos C.; Forbes, Alastair (2011). "Was citrulline first a laxative substance? The truth about modern citrulline and its isolation" (PDF). Nihon Ishigaku Zasshi [Journal of the Japanese Society for the History of Medicine]. 57 (3): 275–292. PMID 22397107.
- ^ Wada, Mitsunori (1930). "On the occurrence of a new amino acid in watermelon. Citrullus vulgaris, Schrad". Proceedings of the Imperial Academy. 6 (1): 15–17. doi:10.2183/pjab1912.6.15.
- ^ Wada, Mitsunori (1930). "On the occurrence of a new amino acid in watermelon, Citrullus vulgaris, Schrad". Bulletin of the Agricultural Chemical Society of Japan. 6 (1–5): 32–34. doi:10.1271/bbb1924.6.32.
- ^ Wada, Mitsunori (1930). "Über Citrullin, eine neue Aminosäure im Preßsaft der Wassermelone, Citrullus vulgaris, Schrad". Biochemische Zeitschrift (in German). 224: 420–429.
- ^ "Nos2 - Nitric Oxide Synthase". Uniprot.org. Uniprot Consortium. Retrieved 10 February 2015.
- ^ Cox M, Lehninger AL, Nelson DR (2000). Lehninger principles of biochemistry (3rd ed.). New York: Worth Publishers. p. 449. ISBN 978-1-57259-153-0. Retrieved 13 March 2020.
- ^ Rogers, G. E.; Rothnagel, J. A. (1983). "A sensitive assay for the enzyme activity in hair follicles and epidermis that catalyses the peptidyl-arginine-citrulline post-translational modification". Current Problems in Dermatology. 11: 171–184. doi:10.1159/000408673. ISBN 978-3-8055-3752-0. PMID 6653155.
- ^ DeLegge, Mark H. (2019-01-01), Corrigan, Mandy L.; Roberts, Kristen; Steiger, Ezra (eds.), "Chapter 7 - Enteral Access and Enteral Nutrition in Patients With Short Bowel Syndrome", Adult Short Bowel Syndrome, Academic Press, pp. 81–96, doi:10.1016/b978-0-12-814330-8.00007-x, ISBN 978-0-12-814330-8, S2CID 81295089, retrieved 2020-11-10
- ^ "Saccharomyces cerevisiae citrulline biosynthesis".
- ^ Fragkos, Konstantinos C.; Forbes, Alastair (2017-10-12). "Citrulline as a marker of intestinal function and absorption in clinical settings: A systematic review and meta-analysis". United European Gastroenterology Journal. 6 (2): 181–191. doi:10.1177/2050640617737632. PMC 5833233. PMID 29511548.
- ^ Crenn, P.; et al. (2000). "Post-absorptive plasma citrulline concentration is a marker of intestinal failure in short bowel syndrome patients". Gastroenterology. 119 (6): 1496–505. doi:10.1053/gast.2000.20227. PMID 11113071.
- ^ Miller, Kelli. "L-citrulline". WebMD. WebMD LLC. Retrieved 16 December 2024.
- ^ Label on Reign Inferno Watermelon Overlord can; Reign Beverage Company: Los Angeles, 2020
