Haloarchaea
| Haloarchaea | |
|---|---|
| |
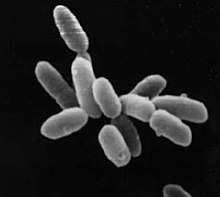
| Halobacterium sp. strain NRC-1, each cell about 5 µm in length. | |
| Scientific classification | |
| Domain: | |
| Kingdom: | |
| Phylum: | |
| Class: | Halobacteria Grant et al. 2002
|
| Order | |
| Synonyms | |
| |
Haloarchaea (halophilic archaea, halophilic archaebacteria, halobacteria)[1] are a class of prokaryotic archaea under the phylum Euryarchaeota,[2] found in water saturated or nearly saturated with salt. 'Halobacteria' are now recognized as archaea rather than bacteria and are one of the largest groups or archaea. The name 'halobacteria' was assigned to this group of organisms before the existence of the domain Archaea was realized, and while valid according to taxonomic rules, should be updated.[3] Halophilic archaea are generally referred to as haloarchaea to distinguish them from halophilic bacteria.
These halophilic microorganisms require high salt concentrations to grow, with most species requiring more than 2M NaCl for growth and survival.[4] They are a distinct evolutionary branch of the Archaea distinguished by the possession of ether-linked lipids and the absence of murein in their cell walls.
Haloarchaea can grow aerobically or anaerobically. Parts of the membranes of haloarchaea are purplish in color,[5] and large blooms of haloarchaea appear reddish from the pigment bacteriorhodopsin, related to the retinal pigment rhodopsin, which it uses to transform light energy into chemical energy by a process unrelated to chlorophyll-based photosynthesis.
Haloarchaea have a potential to solubilize phosphorus. Phosphorus-solubilizing halophilic archaea may well play a role in making phosphorus available to vegetation growing in hypersaline soils. Haloarchaea may also have applications as inoculants for crops growing in hypersaline regions.[6]
Taxonomy
[edit]The extremely halophilic, aerobic members of Archaea are classified within the family Halobacteriaceae, order Halobacteriales in Class III. Halobacteria of the phylum Euryarchaeota (International Committee on Systematics of Prokaryotes, Subcommittee on the taxonomy of Halobacteriaceae). As of May 2016, the family Halobacteriaceae comprises 213 species in 50 genera.
Gupta et al.[7][8] divides the class of Halobacteria in three orders.
- Halobacteriales Grant and Larsen 1989
- Haloarculaceae Gupta et al. 2016, 10 genera
- Halobacteriaceae Gibbons 1974, 24 genera
- Halococcaceae Gupta et al. 2016, 1 genus
- Haloferacales Gupta et al. 2015
- Haloferacaceae Gupta et al. 2015, 10 genera
- Halorubraceae Gupta et al. 2016, 9 genera
- Natrialbales Gupta et al. 2015
- Natrialbaceae Gupta et al. 2015, 18 genera
Phylogeny
[edit]The currently accepted taxonomy is based on the List of Prokaryotic names with Standing in Nomenclature (LPSN)[9] and National Center for Biotechnology Information (NCBI).[2]
| 16S rRNA based LTP_06_2022[10][11][12] | 53 marker proteins based GTDB 08-RS214[13][14][15] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
Note: * paraphyletic Halobacteriaceae
Molecular signatures
[edit]Detailed phylogenetic and comparative analyses of genome sequences from members of the class Haloarchaea has led to division of this class into three orders, Halobacteriales, Haloferacales and Natrialbales, which can be reliably distinguished from each other as well as all other archaea/bacteria through molecular signatures known as conserved signature indels (CSIs).[7] These studies have also identified 68 conserved signature proteins (CSPs) whose homologs are only found in the members of these three orders and 13 CSIs in different proteins that are uniquely present in the members of the class Haloarchaea.[7] These CSIs are present in the following proteins: DNA topoisomerase VI, nucleotide sugar dehydrogenase, ribosomal protein L10e, RecJ-like exonuclease, ribosomal protein S15, adenylosuccinate synthase, phosphopyruvate hydratase, RNA-associated protein, threonine synthase, aspartate aminotransferase, precorrin-8x methylmutase, protoporphyrin IX magnesium chelatase and geranylgeranylglyceryl phosphate synthase-like protein.[7]
Living environment
[edit]
Haloarchaea require salt concentrations in excess of 2 mol/L (or about 10%, three times the ocean salinity which is around 35g/L salt – 3.5%) in the water to grow, and optimal growth usually occurs at much higher concentrations, typically 20–30% (3.4 - 5.2 mol/L of NaCl). [16] However, Haloarchaea can grow up to saturation (about 37% salts).[17] Optimal growth also occurs when pH is neutral or basic and at 45°C temperature. Some haloarchaea can grow even when temperatures exceed 50°C. [16]
Haloarchaea are found mainly in hypersaline lakes and solar salterns. Their high densities in the water often lead to pink or red colourations of the water (the cells possessing high levels of carotenoid pigments, presumably for UV protection).[18] The pigmentation will become enhanced when oxygen levels are low due to an increase in a red pigmented ATP. [16] Some of them live in underground rock salt deposits, including one from middle-late Eocene (38-41 million years ago).[19] Some even older ones from more than 250 million years ago have been reported.[20] Haloarchaea are also used to treat water high in salinity. This is due to its ability to withstand high nutrient levels and the heavy metals that may be present.[16]
Adaptations to environment
[edit]Haloarchaea can grow at water activity (aw) close to 0.75, even though aw lower than 0.90 is inhibitory to most microbes.[21] The high solute concentration causes osmotic stress on microbes, which can cause cell lysis, unfolding of proteins, and inactivation of enzymes.[22] Haloarchaea combat this by retaining compatible solutes such as potassium chloride (KCl) in their intracellular space to allow them to balance osmotic pressure.[23] Retaining these salts is referred to as the “salt-in” method where the cell accumulates a high internal concentration of potassium.[24] Because of the elevated potassium levels, haloarchaea have specialized proteins that have a highly negative surface charge to tolerate high potassium concentrations.[25]
Haloarchaea have adapted to use glycerol as a carbon and energy source in catabolic processes, which is often present in high salt environments due to Dunaliella species that produce glycerol in large quantities.[24]
Phototrophy
[edit]Bacteriorhodopsin is used to absorb light, which provides energy to transport protons (H+) across the cellular membrane. The concentration gradient generated from this process can then be used to synthesize ATP. Many haloarchaea also possess related pigments, including halorhodopsin, which pump chloride ions in the cell in response to photons, creating a voltage gradient and assisting in the production of energy from light. The process is unrelated to other forms of photosynthesis involving electron transport, however, and haloarchaea are incapable of fixing carbon from carbon dioxide.[26] Early evolution of retinal proteins has been proposed in the purple Earth hypothesis.[5]
Cellular shapes
[edit]Haloarchaea are often considered pleomorphic, or able to take on a range of shapes—even within a single species. This makes identification by microscopic means difficult, and it is now more common to use gene sequencing techniques for identification instead.
One of the more unusually shaped Haloarchaea is the "Square Haloarchaeon of Walsby", classified in 2004 using a very low nutrition solution to allow growth along with a high salt concentration. Haloquadratum is square in shape and extremely thin (like a postage stamp). This shape is probably only permitted by the high osmolarity of the water, permitting cell shapes that would be difficult, if not impossible, under other conditions.
As exophiles
[edit]Haloarchaea have been proposed as a kind of life that could live on Mars; since the Martian atmosphere has a pressure below the triple point of water, freshwater species would have no habitat on the Martian surface. The presence of high salt concentrations in water lowers its freezing point, in theory allowing for halophiles to exist in saltwater on Mars.[27] Recently, haloarchaea were sent 36 km (about 22 miles) up into Earth's atmosphere, within a balloon. The two types that were sent up were able to survive the freezing temperatures and high radiation levels, supporting the hypothesis that halophiles could survive on Mars.[28]
Medical use
[edit]Certain types of haloarchaea that produce carotenoids could potentially serve as a source of carotenoids for medical use.[29] Haloarchaea have been proposed to help meet the high demand of carotenoids by pharmaceutical companies due to how easy it can be grown in a lab.[30] Genes in Haloarchaea can also be manipulated in order to produce various strands of carotenoids, further helping meet pharmaceutical companies' needs.[29]
Haloarchaea are also present within the human gut, mostly predominant in the gut of people who live in Korea. Haloarchaea are most abundant in Koreans' guts rather than methanogens due to their saltier diets. This also shows that the archaeome in the human gut can vary drastically depending on region and what is eaten.[31]
Climate change
[edit]Certain types of haloarchaea have been proposed as sources of biodegradable plastics, which could help decrease plastic pollution. Haloarchaea are able to produce polyhydroxyalkanote (PHA), polyhydroxybutyrate (PHB), and polyhydroxyvalerate (PHV) when exposed to certain conditions. For large-scale production of these bioplastics, haloarchaea are favored due to the low cost, fast growth, and lack of need to sterilize area due to the salty environment they prefer. They are also a cleaner option for bioplastics due to them not needing chemicals for cell lysis and the higher recyclability of the process.[32]
Certain types of haloarchaea have also been found to have denitrifying characteristics. If haloarchaea are complete denitrifiers, they could aid salt marshes and other salty environments by buffering these areas of nitrate and nitrite. This could help animal diversity and decrease pollution in these waterways. However, when tested in the lab, haloarchaea have been found to be partial denitrifiers. This means that if haloarchaea are used to treat areas that are high in nitrite and nitrate, they could contribute to nitrogen contaminates and cause an increase in ozone depletion, furthering climate change.[33] The only type of haloarchaea that has been found to reduce atmospheric nitrogen pollution is Haloferax mediterranei.[34] This shows that haloarchaea may be contributing to nitrogen pollution and are not a suitable solution for reducing nitrate and nitrite within high-salinity areas.
See also
[edit]References
[edit]- ^ Fendrihan S, Legat A, Pfaffenhuemer M, Gruber C, Weidler G, Gerbl F, Stan-Lotter H (August 2006). "Extremely halophilic archaea and the issue of long-term microbial survival". Re/Views in Environmental Science and Bio/Technology. 5 (2–3): 203–218. doi:10.1007/s11157-006-0007-y. PMC 3188376. PMID 21984879.
- ^ a b Sayers; et al. "Halobacteria". National Center for Biotechnology Information (NCBI) taxonomy database. Retrieved 2022-06-05.
- ^ DasSarma P, DasSarma S (May 2008). "On the origin of prokaryotic "species": the taxonomy of halophilic Archaea". Saline Systems. 4 (1): 5. doi:10.1186/1746-1448-4-5. PMC 2397426. PMID 18485204.
- ^ DasSarma S, DasSarma P (2017). "Halophiles". eLS. John Wiley & Sons, Ltd. pp. 1–13. doi:10.1002/9780470015902.a0000394.pub4. ISBN 9780470015902.
- ^ a b DasSarma S, Schwieterman EW (2018). "Early evolution of purple retinal pigments on Earth and implications for exoplanet biosignatures". International Journal of Astrobiology. 20 (3): 241–250. arXiv:1810.05150. doi:10.1017/S1473550418000423. ISSN 1473-5504. S2CID 119341330.
- ^ Yadav AN, Sharma D, Gulati S, Singh S, Dey R, Pal KK, et al. (July 2015). "Haloarchaea Endowed with Phosphorus Solubilization Attribute Implicated in Phosphorus Cycle". Scientific Reports. 5: 12293. Bibcode:2015NatSR...512293Y. doi:10.1038/srep12293. PMC 4516986. PMID 26216440.
- ^ a b c d Gupta RS, Naushad S, Baker S (March 2015). "Phylogenomic analyses and molecular signatures for the class Halobacteria and its two major clades: a proposal for division of the class Halobacteria into an emended order Halobacteriales and two new orders, Haloferacales ord. nov. and Natrialbales ord. nov., containing the novel families Haloferacaceae fam. nov. and Natrialbaceae fam. nov". International Journal of Systematic and Evolutionary Microbiology. 65 (Pt 3): 1050–1069. doi:10.1099/ijs.0.070136-0. PMID 25428416.
- ^ Gupta RS, Naushad S, Fabros R, Adeolu M (April 2016). "A phylogenomic reappraisal of family-level divisions within the class Halobacteria: proposal to divide the order Halobacteriales into the families Halobacteriaceae, Haloarculaceae fam. nov., and Halococcaceae fam. nov., and the order Haloferacales into the families, Haloferacaceae and Halorubraceae fam nov". Antonie van Leeuwenhoek. 109 (4): 565–587. doi:10.1007/s10482-016-0660-2. PMID 26837779. S2CID 10437481.
- ^ J.P. Euzéby. "Halobacteria". List of Prokaryotic names with Standing in Nomenclature (LPSN). Retrieved 2021-11-17.
- ^ "The LTP". Retrieved 10 May 2023.
- ^ "LTP_all tree in newick format". Retrieved 10 May 2023.
- ^ "LTP_06_2022 Release Notes" (PDF). Retrieved 10 May 2023.
- ^ "GTDB release 08-RS214". Genome Taxonomy Database. Retrieved 10 May 2023.
- ^ "ar53_r214.sp_label". Genome Taxonomy Database. Retrieved 10 May 2023.
- ^ "Taxon History". Genome Taxonomy Database. Retrieved 10 May 2023.
- ^ a b c d Li J, Gao Y, Dong H, Sheng GP (February 2022). "Haloarchaea, excellent candidates for removing pollutants from hypersaline wastewater". Trends in Biotechnology. 40 (2): 226–239. doi:10.1016/j.tibtech.2021.06.006. PMID 34284891. S2CID 236158869.
- ^ Yadav AN, Sharma D, Gulati S, Singh S, Dey R, Pal KK, et al. (July 2015). "Haloarchaea Endowed with Phosphorus Solubilization Attribute Implicated in Phosphorus Cycle". Scientific Reports. 5: 12293. Bibcode:2015NatSR...512293Y. doi:10.1038/srep12293. PMC 4516986. PMID 26216440.
- ^ DasSarma S (2007). "Extreme Microbes". American Scientist. 95 (3): 224–231. doi:10.1511/2007.65.1024. ISSN 0003-0996.
- ^ Jaakkola ST, Zerulla K, Guo Q, Liu Y, Ma H, Yang C, et al. (2014). "Halophilic archaea cultivated from surface sterilized middle-late eocene rock salt are polyploid". PLOS ONE. 9 (10): e110533. Bibcode:2014PLoSO...9k0533J. doi:10.1371/journal.pone.0110533. PMC 4206341. PMID 25338080.
- ^ Vreeland RH, Rosenzweig WD, Lowenstein T, Satterfield C, Ventosa A (February 2006). "Fatty acid and DNA analyses of Permian bacteria isolated from ancient salt crystals reveal differences with their modern relatives". Extremophiles. 10 (1): 71–78. doi:10.1007/s00792-005-0474-z. PMID 16133658. S2CID 25102006.
- ^ Stevenson A, Cray JA, Williams JP, Santos R, Sahay R, Neuenkirchen N, et al. (June 2015). "Is there a common water-activity limit for the three domains of life?". The ISME Journal. 9 (6): 1333–1351. doi:10.1038/ismej.2014.219. PMC 4438321. PMID 25500507.
- ^ Cheftel JC (1 August 1995). "Review : High-pressure, microbial inactivation and food preservation". Food Science and Technology International. 1 (2–3): 75–90. doi:10.1177/108201329500100203. S2CID 85703396.
- ^ da Costa MS, Santos H, Galinski EA (1998). Biotechnology of Extremophiles. Advances in Biochemical Engineering/Biotechnology. Vol. 61. Springer, Berlin, Heidelberg. pp. 117–153. doi:10.1007/bfb0102291. ISBN 978-3-540-63817-9. PMID 9670799.
- ^ a b Williams TJ, Allen M, Tschitschko B, Cavicchioli R (March 2017). "Glycerol metabolism of haloarchaea". Environmental Microbiology. 19 (3): 864–877. doi:10.1111/1462-2920.13580. hdl:1959.4/unsworks_49888. PMID 27768817.
- ^ Soppa J, Baumann A, Brenneis M, Dambeck M, Hering O, Lange C (September 2008). "Genomics and functional genomics with haloarchaea". Archives of Microbiology. 190 (3): 197–215. doi:10.1007/s00203-008-0376-4. PMID 18493745. S2CID 21222667.
- ^ Bryant DA, Frigaard NU (November 2006). "Prokaryotic photosynthesis and phototrophy illuminated". Trends in Microbiology. 14 (11): 488–496. doi:10.1016/j.tim.2006.09.001. PMID 16997562.
- ^ DasSarma S (2006). "Extreme halophiles are models for astrobiology" (PDF). Microbe-American Society for Microbiology. 1 (3): 120. Archived from the original (PDF) on 2007-02-02.
- ^ DasSarma P, DasSarma S (June 2018). "Survival of microbes in Earth's stratosphere". Current Opinion in Microbiology. Environmental Microbiology * The New Microscopy. 43: 24–30. doi:10.1016/j.mib.2017.11.002. PMID 29156444. S2CID 19041112.
- ^ a b Giani M, Miralles-Robledillo JM, Peiró G, Pire C, Martínez-Espinosa RM (March 2020). "Deciphering Pathways for Carotenogenesis in Haloarchaea". Molecules. 25 (5): 1197. doi:10.3390/molecules25051197. PMC 7179442. PMID 32155882.
- ^ Rodrigo-Baños M, Montero Z, Torregrosa-Crespo J, Garbayo I, Vílchez C, Martínez-Espinosa RM (2021). "Haloarchaea: A Promising Biosource for Carotenoid Production". In Misawa N (ed.). Carotenoids: Biosynthetic and Biofunctional Approaches. Advances in Experimental Medicine and Biology. Vol. 1261. Singapore: Springer. pp. 165–174. doi:10.1007/978-981-15-7360-6_13. ISBN 978-981-15-7360-6. PMID 33783738. S2CID 232419066.
- ^ Kim JY, Whon TW, Lim MY, Kim YB, Kim N, Kwon MS, et al. (August 2020). "The human gut archaeome: identification of diverse haloarchaea in Korean subjects". Microbiome. 8 (1): 114. doi:10.1186/s40168-020-00894-x. PMC 7409454. PMID 32753050.
- ^ Simó-Cabrera L, García-Chumillas S, Hagagy N, Saddiq A, Tag H, Selim S, et al. (March 2021). "Haloarchaea as Cell Factories to Produce Bioplastics". Marine Drugs. 19 (3): 159. doi:10.3390/md19030159. PMC 8003077. PMID 33803653.
- ^ Torregrosa-Crespo J, Bergaust L, Pire C, Martínez-Espinosa RM (February 2018). "Denitrifying haloarchaea: sources and sinks of nitrogenous gases". FEMS Microbiology Letters. 365 (3). doi:10.1093/femsle/fnx270. hdl:10045/73332. PMID 29237000.
- ^ Torregrosa-Crespo J, Pire C, Martínez-Espinosa RM, Bergaust L (January 2019). "Denitrifying haloarchaea within the genus Haloferax display divergent respiratory phenotypes, with implications for their release of nitrogenous gases". Environmental Microbiology. 21 (1): 427–436. doi:10.1111/1462-2920.14474. hdl:10045/83647. PMID 30421557. S2CID 53292259.
Further reading
[edit]Journals
[edit]- Soppa J (March 2006). "From genomes to function: haloarchaea as model organisms". Microbiology. 152 (Pt 3): 585–590. doi:10.1099/mic.0.28504-0. PMID 16514139.
- Cavalier-Smith T (January 2002). "The neomuran origin of archaebacteria, the negibacterial root of the universal tree and bacterial megaclassification". International Journal of Systematic and Evolutionary Microbiology. 52 (Pt 1): 7–76. doi:10.1099/00207713-52-1-7. PMID 11837318.
- Woese CR, Kandler O, Wheelis ML (June 1990). "Towards a natural system of organisms: proposal for the domains Archaea, Bacteria, and Eucarya". Proceedings of the National Academy of Sciences of the United States of America. 87 (12): 4576–4579. Bibcode:1990PNAS...87.4576W. doi:10.1073/pnas.87.12.4576. PMC 54159. PMID 2112744.
- Cavalier-Smith T (1986). "The kingdoms of organisms". Nature. 324 (6096): 416–417. Bibcode:1986Natur.324..416C. doi:10.1038/324416a0. PMID 2431320. S2CID 5242667.
Books
[edit]- Grant WD, Kamekura M, McGenity TJ, Ventosa A (2001). "Class III. Halobacteria class. nov.". In DR Boone, RW Castenholz (eds.). Bergey's Manual of Systematic Bacteriology Volume 1: The Archaea and the deeply branching and phototrophic Bacteria (2nd ed.). New York: Springer Verlag. pp. 169. ISBN 978-0-387-98771-2.
- Garrity GM, Holt JG (2001). "Phylum AII. Euryarchaeota phy. nov.". In DR Boone, RW Castenholz (eds.). Bergey's Manual of Systematic Bacteriology Volume 1: The Archaea and the deeply branching and phototrophic Bacteria (2nd ed.). New York: Springer Verlag. pp. 169. ISBN 978-0-387-98771-2.
