Ff phages

Ff phages (for F specific filamentous phages) is a group of almost identical filamentous phage (genus Inovirus) including phages f1, fd, M13 and ZJ/2, which infect bacteria bearing the F fertility factor.[1][2][3][4][5][6][7] The virion (virus particle) is a flexible filament measuring about 6 by 900 nm, comprising a cylindrical protein tube protecting a single-stranded circular DNA molecule at its core. The phage codes for only 11 gene products, and is one of the simplest viruses known. It has been widely used to study fundamental aspects of molecular biology. George Smith and Greg Winter used f1 and fd for their work on phage display for which they were awarded a share of the 2018 Nobel Prize in Chemistry.[8] Early experiments on Ff phages used M13 to identify gene functions,[9][10] and M13 was also developed as a cloning vehicle,[11] so the name M13 is sometimes used as an informal synonym for the whole group of Ff phages.
Structure
[edit]
The virion is a flexible filament (worm-like chain) about 6 nm in diameter and 900 nm long. Several thousand copies of a small (50 amino-acid residues) elongated alpha-helical major coat protein subunit (the product of gene 8, or p8) in an overlapping shingle-like array form a hollow cylinder enclosing the circular single-stranded DNA genome. Each p8 subunit has a collection of basic residues near the C-terminus of the elongated protein and acidic residues near the N-terminus; these two regions are separated by about 20 hydrophobic (non-polar) residues. The shingle-like arrangement places the acidic residues of p8 near the outside surface of the cylinder, where they cause the virus particle to be negatively-charged; non-polar regions near non-polar regions of neighbouring p8 subunits, where non-polar interactions contribute to a notable physical stability of the virus particle; and basic residues near the centre of the cylinder, where they interact with the negatively-charged DNA phosphates at the core of the virion. Longer[12] (or shorter[13]) DNA molecules can be packaged, since more (or fewer) p8 subunits can be added during assembly as required to protect the DNA, making the phage useful for genetic studies. (This effect should not be confused with polyphage, which can package several separate and distinct DNA molecules). About 5 copies each of four minor proteins cap the two ends of the virion.[14]
The molecular structure of the virion capsid (the assembly of p8 subunit proteins) has been determined by X-ray fiber diffraction, and structural models have been deposited in the Protein Data Bank. In particular, the series of fd and Pf1 virion structures deposited in the PDB over decades illustrate the improvements in methods for fiber diffraction data collection and computational analysis. Structures of the p3 capsid protein and the p5 replication/assembly protein have also been determined from X-ray crystallography and deposited in the PDB.[citation needed]
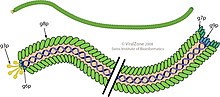
Genetics
[edit]The DNA sequence of the fd genome has 6408 nucleotide comprising 9 genes, but the genome has 11 open reading frames producing 11 proteins, since two genes, gene 2 and gene 1, have internal in-frame translation starts, generating two additional proteins, p10 and p11. The genome also contains a short non-coding intergenic sequence.[15] M13 and f1 sequences are slightly different from fd. They both have only 6407 nucleotides; f1 differs from fd in 180 positions (only 10 of these changes are reflected in amino-acid changes in gene products)[16] and M13 has only 59 nucleotide differences from f1. For many purposes the phages in the Ff group can be considered as interchangeable.
Five gene products are part of the virion: the major coat protein (p8) and the minor proteins capping the two ends, p3 and p6 at one end, and p7 and p9 at the other end. Three gene products (p2, p5, and p10) are cytoplasmic proteins needed for DNA synthesis and the rest are membrane proteins involved in assembly of the virion.[17]
| Inovirus | |
|---|---|
| Virus classification | |
| (unranked): | Virus |
| Realm: | Monodnaviria |
| Kingdom: | Loebvirae |
| Phylum: | Hofneiviricota |
| Class: | Faserviricetes |
| Order: | Tubulavirales |
| Family: | Inoviridae |
| Genus: | Inovirus |
The gene encoding p1 has been used as a conserved marker gene, along with three other features specific for inovirus genomes, in an automatic machine-learning approach to identify over 10000 inovirus-like sequences from microbial genomes.[18]
Replication cycle
[edit]Infection
[edit]The p3 protein is anchored to one end of the virion by the C-terminal domain of p3. Infection of host bacteria involves interaction of two different N-terminal regions of p3 with two different sites of the host bacteria. First, the N2 domain of p3 attaches to the outer tip of the F-pilus, and the pilus retracts into the cell. This retraction may involve depolymerization of the pilus subunit assembly into the cell membrane at the base of the pilus by a reversal of the pilus growth and polymerization process.[1][19][20] As the tip of the pilus bearing p3 approaches the cell wall, the N1 domain of p3 interacts with the bacterial TolQRA protein to complete infection and release the genome into the cytoplasm of the host.[21][22]
Replication
[edit]After the single-stranded viral DNA enters the cytoplasm, it serves as a template for the synthesis of a complementary DNA strand. This synthesis is initiated in the intergenic region of the DNA sequence by host RNA polymerase, which synthesizes a short RNA primer on the infecting DNA as template. The host DNA polymerase III then uses this primer to synthesize the full complementary strand of DNA, yielding a double-stranded circle, sometimes called the replicative form (RF) DNA. The complementary strand of the RF is the transcription template for phage coded proteins, especially p2 and p10, which are necessary for further DNA replication.[citation needed]
The p2 protein cleaves the viral strand of the RF DNA, and host DNA polymerase III synthesizes a new viral strand. The old viral strand is displaced as the new one is synthesized. When a circle is complete, the covalently linked p2 cuts the displaced viral strand at the junction between the old and newly synthesized DNA and re-ligates the two ends and liberates p2. RF replicates by this rolling circle mechanism to generate dozens of copies of the RF.[citation needed]
When the concentration of phage proteins has increased, new viral strands are coated by the replication/assembly protein p5 rather than by the complementary DNA strands. The p5 also inhibits translation of p2, so that progeny viral ssDNA production and packaging are in synchrony.[6]
Assembly and extrusion
[edit]Infection does not kill the host bacteria,[23] in contrast to most other families of phage. Progeny phage are assembled as they extrude through the membrane of growing bacteria, probably at adhesion sites joining inner and outer membranes. The five phage proteins that form the coat of the completed phage enter the inner membrane; for p8 and p3, N-terminal leader sequences (later removed) help the proteins to enter the bacterial membrane, with their N-termini directed away from the cytoplasm towards the periplasm. Three other phage membrane proteins that are not present in the phage, p1, p11, and p4, are also involved in assembly. Replication of RF DNA is converted to production of phage ssDNA by coating of the DNA with p5 to form an elongated p5/DNA replication/assembly complex, which then interacts with the membrane-bound phage proteins. The extrusion process picks up the p7 and p9 proteins which form the outer tip of the progeny phage. As the p5 is stripped off the DNA, the progeny DNA is extruded across the membrane and wrapped in a helical casing of p8, to which p3 and p6 are added at the end of assembly. The p4 protein may form an extrusion pore in the outer membrane.[14]
Interaction of the double-stranded packaging DNA signal with the p1-thioredoxin complex at the host inner membrane triggers the formation of a pore. The p1 protein contains Walker motifs which are essential for phage assembly,[24] suggesting that p1 is a molecular motor involved in phage assembly. The p1 protein has a membrane-spanning hydrophobic domain with the N-terminal portion in the cytoplasm and the C-terminal portion in the periplasm (the reverse of the orientation of p8). Adjacent to the cytoplasmic side of the membrane-spanning domain is a 13- residue sequence of p1 having a pattern of basic residues closely matching the pattern of basic residues near the C terminus of p8, but inverted with respect to that sequence.[25]
Intermediate assemblies of p8 can be generated by treating the phage with chloroform.[26][27][28] The helical content of p8 in these intermediate forms is similar to that in the phage, suggesting that the structural change during assembly may involve just a sliding of the shingled p8 subunits with respect to their neighbours in the assembly.[29][30]
Applications
[edit]Life sciences and medicine
[edit]Ff phages have been engineered for applications in biological and medical sciences. Many applications build on experiments[12] showing that the DNA sequence determining resistance to the antibiotic kanamycin can be inserted in a functional form into the non-coding intergenic sequence of fd phage DNA. Such modified phage are correspondingly longer that wild-type filamentous fd, because the longer DNA is coated with correspondingly more gene 8 coat proteins, but the phage life-cycle is not otherwise disrupted. The traditional “tadpole” or isometric shaped-phage, on the other hand, which have a limited-sized capsid, cannot be so easily used to encapsidate a larger DNA molecule. The modified phage can be selected by infecting kanamycin-sensitive bacteria with modified phage to introduce resistance to kanamycin, and growing the infected bacteria in media containing an otherwise lethal concentration of kanamycin.[citation needed]
This result was extended by inserting foreign DNA expressing a foreign peptide into fd phage gene 3, rather than into the intergenic sequence, so that the foreign peptide appears on the surface of the phage as a part of the gene 3 adsorption protein.[31][32][33] Phage carrying the foreign peptide can then be detected using appropriate antibodies. The reverse of this approach is to insert DNA coding for antibodies into gene 3 and detect their presence by appropriate antigens.[34]
These techniques have been extended over the years in many ways, for instance by inserting foreign DNA into the genes coding for phage coat proteins other than gene 3, and/or duplicating the gene of interest to modify only some of the corresponding gene products. Phage display technology has been widely used for many purposes. [35][36][37]
Material sciences and nanotechnology
[edit]Ff phages have been engineered for applications such as remediation, electrochemical, photovoltaic, catalytic, sensing and digital memory devices, especially by Angela Belcher and colleagues.[6][38][39][40][41][42][43][44][45][excessive citations]
See also
[edit]References
[edit]- ^ a b Rasched I, Oberer E (December 1986). "Ff coliphages: structural and functional relationships". Microbiological Reviews. 50 (4): 401–27. doi:10.1128/MR.50.4.401-427.1986. PMC 373080. PMID 3540571.
- ^ Mai-Prochnow A, Hui JG, Kjelleberg S, Rakonjac J, McDougald D, Rice SA (July 2015). "'Big things in small packages: the genetics of filamentous phage and effects on fitness of their host'". FEMS Microbiology Reviews. 39 (4): 465–87. doi:10.1093/femsre/fuu007. hdl:10453/65260. PMID 25670735.
- ^ Rakonjac J, Bas B, Derda R, eds. (2017). Filamentous Bacteriophage in Bio/Nano/Technology, Bacterial Pathogenesis and Ecology. Frontiers Research Topics. Frontiers Media SA. doi:10.3389/978-2-88945-095-4. ISBN 978-2-88945-095-4.
- ^ Morag O, Abramov G, Goldbourt A (December 2011). "Similarities and differences within members of the Ff family of filamentous bacteriophage viruses". The Journal of Physical Chemistry B. 115 (51): 15370–9. doi:10.1021/jp2079742. PMID 22085310.
- ^ Hay ID, Lithgow T (June 2019). "Filamentous phages: masters of a microbial sharing economy". EMBO Reports. 20 (6). doi:10.15252/embr.201847427. PMC 6549030. PMID 30952693.
- ^ a b c Rakonjac J, Bennett NJ, Spagnuolo J, Gagic D, Russel M (2011). "Filamentous bacteriophage: biology, phage display and nanotechnology applications". Current Issues in Molecular Biology. 13 (2): 51–76. PMID 21502666.
- ^ Rakonjac J, Russel M, Khanum S, Brooke SJ, Rajič M (2017). "Filamentous Phage: Structure and Biology". In Lim TS (ed.). Recombinant Antibodies for Infectious Diseases. Advances in Experimental Medicine and Biology. Vol. 1053. Cham: Springer International Publishing. pp. 1–20. doi:10.1007/978-3-319-72077-7_1. ISBN 978-3-319-72076-0. PMID 29549632.
- ^ "The Nobel Prize in Chemistry 2018". NobelPrize.org. Retrieved 2021-04-10.
- ^ Pratt D, Tzagoloff H, Erdahl WS (November 1966). "Conditional lethal mutants of the small filamentous coliphage M13. I. Isolation, complementation, cell killing, time of cistron action". Virology. 30 (3): 397–410. doi:10.1016/0042-6822(66)90118-8. PMID 5921643.
- ^ Pratt D, Tzagoloff H, Beaudoin J (September 1969). "Conditional lethal mutants of the small filamentous coliphage M13. II. Two genes for coat proteins". Virology. 39 (1): 42–53. doi:10.1016/0042-6822(69)90346-8. PMID 5807970.
- ^ Messing J (April 1991). "Cloning in M13 phage or how to use biology at its best". Gene. 100: 3–12. doi:10.1016/0378-1119(91)90344-b. PMID 2055478.
- ^ a b Herrmann R, Neugebauer K, Zentgraf H, Schaller H (February 1978). "Transposition of a DNA sequence determining kanamycin resistance into the single-stranded genome of bacteriophage fd". Molecular & General Genetics. 159 (2): 171–8. doi:10.1007/BF00270890. PMID 345091. S2CID 22923713.
- ^ Sattar S, Bennett NJ, Wen WX, Guthrie JM, Blackwell LF, Conway JF, Rakonjac J (2015). "Ff-nano, short functionalized nanorods derived from Ff (f1, fd, or M13) filamentous bacteriophage". Frontiers in Microbiology. 6: 316. doi:10.3389/fmicb.2015.00316. PMC 4403547. PMID 25941520.
- ^ a b Straus SK, Bo HE (2018). "Filamentous Bacteriophage Proteins and Assembly". Virus Protein and Nucleoprotein Complexes. Subcellular Biochemistry. Vol. 88. pp. 261–279. doi:10.1007/978-981-10-8456-0_12. ISBN 978-981-10-8455-3. PMID 29900501.
- ^ Beck E, Sommer R, Auerswald EA, Kurz C, Zink B, Osterburg G, et al. (December 1978). "Nucleotide sequence of bacteriophage fd DNA". Nucleic Acids Research. 5 (12): 4495–503. doi:10.1093/nar/5.12.4495. PMC 342768. PMID 745987.
- ^ Beck E, Zink B (December 1981). "Nucleotide sequence and genome organisation of filamentous bacteriophages fl and fd". Gene. 16 (1–3): 35–58. doi:10.1016/0378-1119(81)90059-7. PMID 6282703.
- ^ Russel M, Linderoth NA, Sali A (June 1997). "Filamentous phage assembly: variation on a protein export theme". Gene. 192 (1): 23–32. doi:10.1016/s0378-1119(96)00801-3. PMID 9224870.
- ^ Roux S, Krupovic M, Daly RA, Borges AL, Nayfach S, Schulz F, et al. (November 2019). "Cryptic inoviruses revealed as pervasive in bacteria and archaea across Earth's biomes". Nature Microbiology. 4 (11): 1895–1906. doi:10.1038/s41564-019-0510-x. PMC 6813254. PMID 31332386.
- ^ Lawley TD, Klimke WA, Gubbins MJ, Frost LS (July 2003). "F factor conjugation is a true type IV secretion system". FEMS Microbiology Letters. 224 (1): 1–15. doi:10.1016/S0378-1097(03)00430-0. PMID 12855161.
- ^ Craig L, Forest KT, Maier B (July 2019). "Type IV pili: dynamics, biophysics and functional consequences". Nature Reviews. Microbiology. 17 (7): 429–440. doi:10.1038/s41579-019-0195-4. PMID 30988511. S2CID 115153017.
- ^ Bennett NJ, Rakonjac J (February 2006). "Unlocking of the filamentous bacteriophage virion during infection is mediated by the C domain of pIII". Journal of Molecular Biology. 356 (2): 266–73. doi:10.1016/j.jmb.2005.11.069. PMID 16373072.
- ^ Hoffmann-Thoms S, Jakob RP, Schmid FX (April 2014). "Energetic communication between functional sites of the gene-3-protein during infection by phage fd". Journal of Molecular Biology. 426 (8): 1711–22. doi:10.1016/j.jmb.2014.01.002. PMID 24440124.
- ^ Hoffmann Berling H, Maze R (March 1964). "Release of male-specific bacteriophages from surviving host bacteria BACTERIA". Virology. 22 (3): 305–13. doi:10.1016/0042-6822(64)90021-2. PMID 14127828.
- ^ Loh B, Haase M, Mueller L, Kuhn A, Leptihn S (April 2017). "The Transmembrane Morphogenesis Protein gp1 of Filamentous Phages Contains Walker A and Walker B Motifs Essential for Phage Assembly". Viruses. 9 (4): 73. doi:10.3390/v9040073. PMC 5408679. PMID 28397779.
- ^ Rapoza MP, Webster RE (May 1995). "The products of gene I and the overlapping in-frame gene XI are required for filamentous phage assembly". Journal of Molecular Biology. 248 (3): 627–38. doi:10.1006/jmbi.1995.0247. PMID 7752229.
- ^ Griffith J, Manning M, Dunn K (March 1981). "Filamentous bacteriophage contract into hollow spherical particles upon exposure to a chloroform-water interface". Cell. 23 (3): 747–53. doi:10.1016/0092-8674(81)90438-4. PMID 7226228. S2CID 46531024.
- ^ Manning M, Griffith J (January 1985). "Association of M13 I-forms and spheroids with lipid vesicles". Archives of Biochemistry and Biophysics. 236 (1): 297–303. doi:10.1016/0003-9861(85)90629-0. PMID 3966795.
- ^ Stopar D, Spruijt RB, Wolfs CJ, Hemminga MA (July 1998). "Mimicking initial interactions of bacteriophage M13 coat protein disassembly in model membrane systems". Biochemistry. 37 (28): 10181–7. doi:10.1021/bi9718144. PMID 9665724.
- ^ Roberts LM, Dunker AK (October 1993). "Structural changes accompanying chloroform-induced contraction of the filamentous phage fd". Biochemistry. 32 (39): 10479–88. doi:10.1021/bi00090a026. PMID 8399194.
- ^ Xue, Bin; Blocquel, David; Habchi, Johnny; Uversky, Alexey V.; Kurgan, Lukasz; Uversky, Vladimir N.; Longhi, Sonia (2014). "Structural Disorder in Viral Proteins". Chemical Reviews. 114 (13): 6880–6911. doi:10.1021/cr4005692. ISSN 0009-2665. PMID 24823319.
- ^ Smith, G. (1985). "Filamentous fusion phage: novel expression vectors that display cloned antigens on the virion surface". Science. 228 (4705): 1315–1317. Bibcode:1985Sci...228.1315S. doi:10.1126/science.4001944. ISSN 0036-8075. PMID 4001944.
- ^ Parmley, Stephen F.; Smith, George P. (1988). "Antibody-selectable filamentous fd phage vectors: affinity purification of target genes". Gene. 73 (2): 305–318. doi:10.1016/0378-1119(88)90495-7. ISSN 0378-1119. PMID 3149606.
- ^ Webster, R.E., 2001. Filamentous phage biology. In: Barbas III, C.F., Burton, D.R., Scott, J.K., Silverman, G.J. (Eds.), Phage Display: A Laboratory Manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, pp. 1.1-1.37.
- ^ Winter, Greg; Griffiths, Andrew D.; Hawkins, Robert E.; Hoogenboom, Hennie R. (1994). "Making Antibodies by Phage Display Technology". Annual Review of Immunology. 12 (1): 433–455. doi:10.1146/annurev.iy.12.040194.002245. ISSN 0732-0582. PMID 8011287.
- ^ Prisco A, De Berardinis P (2012). "Filamentous bacteriophage fd as an antigen delivery system in vaccination". International Journal of Molecular Sciences. 13 (4): 5179–94. doi:10.3390/ijms13045179. PMC 3344273. PMID 22606037.
- ^ Henry KA, Arbabi-Ghahroudi M, Scott JK (2015). "Beyond phage display: non-traditional applications of the filamentous bacteriophage as a vaccine carrier, therapeutic biologic, and bioconjugation scaffold". Frontiers in Microbiology. 6: 755. doi:10.3389/fmicb.2015.00755. PMC 4523942. PMID 26300850.
- ^ Sioud M (April 2019). "Phage Display Libraries: From Binders to Targeted Drug Delivery and Human Therapeutics". Molecular Biotechnology. 61 (4): 286–303. doi:10.1007/s12033-019-00156-8. PMID 30729435. S2CID 73434013.
- ^ Dogic Z (2016). "Filamentous Phages As a Model System in Soft Matter Physics". Frontiers in Microbiology. 7: 1013. doi:10.3389/fmicb.2016.01013. PMC 4927585. PMID 27446051.
- ^ Oh D, Qi J, Lu YC, Zhang Y, Shao-Horn Y, Belcher AM (2013). "Biologically enhanced cathode design for improved capacity and cycle life for lithium-oxygen batteries". Nature Communications. 4 (1): 2756. Bibcode:2013NatCo...4.2756O. doi:10.1038/ncomms3756. PMC 3930201. PMID 24220635.
- ^ Dorval Courchesne NM, Klug MT, Huang KJ, Weidman MC, Cantú VJ, Chen PY, et al. (2015). "Constructing Multifunctional Virus-Templated Nanoporous Composites for Thin Film Solar Cells: Contributions of Morphology and Optics to Photocurrent Generation". The Journal of Physical Chemistry C. 119 (25): 13987–14000. doi:10.1021/acs.jpcc.5b00295. hdl:1721.1/102981. ISSN 1932-7447.
- ^ Lee SW, Belcher AM (2004). "Virus-Based Fabrication of Micro- and Nanofibers Using Electrospinning". Nano Letters. 4 (3): 387–390. Bibcode:2004NanoL...4..387L. doi:10.1021/nl034911t. ISSN 1530-6984.
- ^ Casey JP, Barbero RJ, Heldman N, Belcher AM (November 2014). "Versatile de novo enzyme activity in capsid proteins from an engineered M13 bacteriophage library". Journal of the American Chemical Society. 136 (47): 16508–14. doi:10.1021/ja506346f. PMID 25343220.
- ^ Zhang G, Wei S, Belcher AM (2018). "Biotemplated Zinc Sulfide Nanofibers as Anode Materials for Sodium-Ion Batteries". ACS Applied Nano Materials. 1 (10): 5631–5639. doi:10.1021/acsanm.8b01254. hdl:1721.1/126086. ISSN 2574-0970. S2CID 104742577.
- ^ Li L, Belcher AM, Loke DK (December 2020). "Simulating selective binding of a biological template to a nanoscale architecture: a core concept of a clamp-based binding-pocket-favored N-terminal-domain assembly". Nanoscale. 12 (47): 24214–24227. doi:10.1039/D0NR07320B. PMID 33289758. S2CID 227950477.
- ^ Brogan AP, Heldman N, Hallett JP, Belcher AM (September 2019). "Thermally robust solvent-free biofluids of M13 bacteriophage engineered for high compatibility with anhydrous ionic liquids". Chemical Communications. 55 (72): 10752–10755. doi:10.1039/C9CC04909F. hdl:1721.1/125988. PMID 31432818. S2CID 201115233.
