1,2-Bis(dimethylphosphino)ethane
Appearance
(Redirected from Dmpe)
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
(Ethane-1,2-diyl)bis(dimethylphosphane) | |
| Other names
DMPE
ethylenebis(dimethylphosphine) 1,2-Bis(dimethylphosphino)ethane | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.155.809 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H16P2 | |
| Molar mass | 150.142 g·mol−1 |
| Density | 0.9 g/mL at 25 °C |
| Boiling point | 180 °C (356 °F; 453 K) |
| Hazards | |
| GHS labelling:[1] | |
 
| |
| Danger | |
| H225, H250, H315, H319, H335 | |
| P210, P222, P231, P233, P240, P241, P242, P243, P261, P264, P264+P265, P271, P280, P302+P335+P334, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P370+P378, P403+P233, P403+P235, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
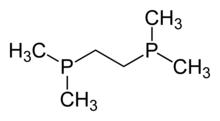
1,2-Bis(dimethylphosphino)ethane (dmpe) is a diphosphine ligand in coordination chemistry. It is a colorless, air-sensitive liquid that is soluble in organic solvents. With the formula (CH2PMe2)2, dmpe is used as a compact strongly basic spectator ligand (Me = methyl), Representative complexes include V(dmpe)2(BH4)2, Mn(dmpe)2(AlH4)2, Tc(dmpe)2(CO)2Cl, and Ni(dmpe)Cl2.[2]

Synthesis
[edit]It is synthesised by the reaction of methylmagnesium iodide with 1,2-bis(dichlorophosphino)ethane:[3]
- Cl2PCH2CH2PCl2 + 4 MeMgI → Me2PCH2CH2PMe2 + 4 MgICl
Alternatively it can be generated by alkylation of sodium dimethylphosphide.
The synthesis of dmpe from thiophosphoryl chloride has led to serious accidents and has been abandoned.[4]
Related ligands
[edit]- Bis(dicyclohexylphosphino)ethane, a bulkier analogue, which is also a solid.
- 1,2-Bis(diphenylphosphino)ethane, more air-stable than dmpe, but less basic.
- 1,2-Bis(dimethylphosphino)benzene, a more rigid analogue of dmpe.
- Tetramethylethylenediamine, the diamine analogue of dmpe.
References
[edit]- ^ Sigma-Aldrich Co., 1,2-Bis(dimethylphosphino)ethane. Retrieved on 2013-07-20.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ R. J. Burt; J. Chatt; W. Hussain; G. J. Leigh (1979). "A convenient synthesis of 1,2-bis(dichlorophosphino)ethane, 1,2-bis(dimethylphosphino)ethane and 1,2-bis(diethylphosphino)ethane". Journal of Organometallic Chemistry. 182 (2): 203–6. doi:10.1016/S0022-328X(00)94383-3.
- ^ Bercaw, J. E.; Parshall, G. W. (1985). "Preparation of Tetramethyldiphosphine Disulfide and Ethylenebis(Dimethylphosphine) (Dmpe)". Inorganic Syntheses. Vol. 23. pp. 199–200. doi:10.1002/9780470132548.ch42. ISBN 9780470132548.
