Balsalazide
 | |
| Clinical data | |
|---|---|
| Trade names | Colazal, Giazo |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a699052 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | <1% |
| Protein binding | ≥99% |
| Elimination half-life | 12hr |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.117.186 |
| Chemical and physical data | |
| Formula | C17H15N3O6 |
| Molar mass | 357.322 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Balsalazide is an anti-inflammatory drug used in the treatment of inflammatory bowel disease. It is sold under the brand names Giazo, Colazal in the US and Colazide in the UK. It is also sold in generic form in the US by several generic manufacturers.
It is usually administered as the disodium salt. Balsalazide releases mesalazine, also known as 5-aminosalicylic acid, or 5-ASA,[2] in the large intestine. Its advantage over that drug in the treatment of ulcerative colitis is believed to be the delivery of the active agent past the small intestine to the large intestine, the active site of ulcerative colitis. It is in the category of disease-modifying antirheumatic drugs (DMARDs) family of medications.[3] It is unclear exactly how it works.[3]
Synthesis
[edit]Ex 3 is actually for Ipsalazide. See Ex 4 for Balsalazide proper. Same protocol but uses β-Alanine.
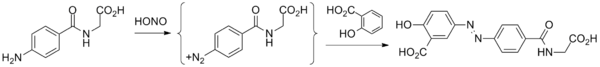
- Starting material is 4-aminohippuric acid, obtained by coupling para-aminobenzoic acid and glycine.
- That product is then treated with nitrous acid to give the diazonium salt.
- Reaction of this species with salicylic acid proceeds at the position para to the phenol to give balsalazide.
References
[edit]- ^ "Colazide 750mg Capsules - Summary of Product Characteristics (SmPC)". (emc). 3 January 2019. Retrieved 2 October 2020.
- ^ Kruis W, Schreiber S, Theuer D, Brandes JW, Schütz E, Howaldt S, et al. (December 2001). "Low dose balsalazide (1.5 g twice daily) and mesalazine (0.5 g three times daily) maintained remission of ulcerative colitis but high dose balsalazide (3.0 g twice daily) was superior in preventing relapses". Gut. 49 (6): 783–9. doi:10.1136/gut.49.6.783. PMC 1728533. PMID 11709512.
- ^ a b "Sulfasalazine". The American Society of Health-System Pharmacists. Archived from the original on 21 December 2016. Retrieved 8 December 2016.
External links
[edit]- "Balsalazide". Drug Information Portal. U.S. National Library of Medicine.
