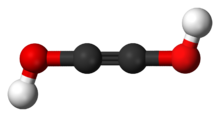
Acetylenediol
| |
| Names | |
|---|---|
| Preferred IUPAC name
Ethynediol | |
| Other names
Dihydroxyacetylene
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2H2O2 | |
| Molar mass | 58.07 g/mol |
| Boiling point | decomposes |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Acetylenediol, or ethynediol, is a chemical substance with formula HO−C≡C−OH (an ynol). It is the diol of acetylene. Acetylenediol is unstable in the condensed phase, although its tautomer glyoxal (CHO)2 is well known.
Detection
[edit]Acetylenediol was first observed in the gas-phase by mass spectrometry.[1] The compound was later obtained by photolysis of squaric acid in a solid argon matrix at 10 K (−263 °C).[2] Recently, this molecule was synthesized in interstellar ice analogs composed of carbon monoxide (CO) and water (H2O) upon exposure to energetic electrons and detected upon sublimation by isomer-selective photoionization reflectron time-of-flight mass spectrometry.[3]
Derivatives
[edit]Alkoxide derivatives
[edit]Like the diol, most simple ether derivatives are labile. Di-tert-butoxyacetylene is however a distillable liquid.
Acetylenediolate salts
[edit]Salts of the acetylenediolate (ethynediolate) dianion −O−C≡C−O− are known. They are not however prepared from ethynediol, but by the reduction of carbon monoxide. Potassium acetylenediolate (K2C2O2) was first obtained by Liebig in 1834, from the reaction of carbon monoxide with metallic potassium;[4] but for a long time the product was assumed to be "potassium carbonyl" (KCO). Over the next 130 years were described the "carbonyls" of sodium (Johannis, 1893), barium (Gunz and Mentrel, 1903), strontium (Roederer, 1906), and lithium, rubidium, and caesium (Pearson, 1933).[5] The reaction was eventually shown to yield a mixture of the potassium acetylenediolate K
2C
2O
2 and potassium benzenehexolate K
6C
6O
6.[6]
The structure of these salts was clarified only in 1963 by Büchner and Weiss.[7][8]
Acetylenediolates can also be prepared by the rapid reaction of CO and a solution of the corresponding metal in liquid ammonia at low temperature.[5] Potassium acetylenediolate is a pale yellow solid that reacts explosively with air, halogens, halogenated hydrocarbons, alcohols, water, and any substance which possesses an acidic hydrogen.[9]
Coordination complexes
[edit]Acetylenediol can form coordination compounds, such as [TaH(HOC≡COH)(dmpe)2Cl]+Cl− where dmpe is bis(dimethylphosphino)ethane.[10]
Acetylenediolate and related anions such as deltate C
3O2−
3 and squarate C
4O2−
4 have been obtained from carbon monoxide under mild conditions by reductive coupling of CO ligands in organouranium complexes.[11]
See also
[edit]References
[edit]- ^ Terlouw, Johan K.; Burgers, Peter C.; van Baar, Ben L. M.; Weiske, Thomas; Schwarz, Helmut (1986). "The Formation in the Gas Phase of HO–CC–OH, H2N–CC–NH2, H2N–CC–OH and Related Compounds by Selective Reduction of Their Cations". CHIMIA. 40: 357–359. Retrieved 1 August 2009.
- ^ Maier, Günther; Rohr, Christine (1995). "Ethynediol: Photochemical generation and matrix-spectroscopic identification". Liebigs Annalen. 1996 (3): 307–309. doi:10.1002/jlac.199619960303.
- ^ Wang, Jia; Turner, Andrew M.; Marks, Joshua H.; Zhang, Chaojiang; Kleimeier, N. Fabian; Bergantini, Alexandre; Singh, Santosh K.; Fortenberry, Ryan C.; Kaiser, Ralf I. (1 June 2024). "Preparation of Acetylenediol (HOCCOH) and Glyoxal (HCOCHO) in Interstellar Analog Ices of Carbon Monoxide and Water". The Astrophysical Journal. 967 (2): 79. doi:10.3847/1538-4357/ad3c3e.
- ^ Justus Liebig (1834), Annalen der Chemie und Pharmacie, volume 11, p. 182. Cited by Raymond N. Vrtis et al. (1988), JACS p. 7564.
- ^ a b T. G. Pearson (1933), Carbonyls of Lithium, Rubidium and Caesium. Nature, volume 131, pp. 166–167 (4 February 1933). doi:10.1038/131166b0
- ^ Werner Büchner, E. Weiss (1964) Zur Kenntnis der sogenannten «Alkalicarbonyle» IV[1] Über die Reaktion von geschmolzenem Kalium mit Kohlenmonoxid. Helvetica Chimica Acta, Volume 47 Issue 6, Pages 1415–1423.doi:10.1002/hlca.19640470604
- ^ Werner Büchner, E. Weiss (1963) Zur Kenntnis der sogenannten «Alkalicarbonyle» I Die Kristallstruktur des Kalium-acetylendiolats, KOC≡COK. Helvetica Chimica Acta, Volume 46 Issue 4, Pages 1121–1127. doi:10.1002/hlca.19630460404
- ^ E. Weiss, Werner Büchner (1963), Zur Kenntnis der sogenannten Alkalicarbonyle. II. Die Kristallstrukturen des Rubidium- und Caesium-acetylendiolats, RbOC≡CORb und CsOC≡COCs. Zeitschrift für anorganische und allgemeine Chemie, Volume 330 Issue 5-6, Pages 251–258. doi:10.1002/zaac.19643300504
- ^ Charles Kenneth Taylor (1982), The Chemical Behavior of the Alkali Metal Acetylenediolates. Thesis, Pennsylvania State University; also Technical Memo A642321, Penn State University Park Applied Research Lab. 227 pages.
- ^ Raymond N. Vrtis, Ch. Pulla Rao, Simon G. Bott, and Stephen J. Lippard (1988), Synthesis and Stabilization of Tantalum-Coordinated Dihydroxyacetylene from Two Reductively Coupled Carbon Monoxide Ligands J. Am. Chem. Soc., volume 110 issue 22, pp. 7564–7566. doi:10.1021/ja00230a062
- ^ Alistair S. Frey, F. Geoffrey N. Cloke, Peter B. Hitchcock (2008), Mechanistic Studies on the Reductive Cyclooligomerisation of CO by U(III) Mixed Sandwich Complexes; the Molecular Structure of [(U(η-C8H6{Si'Pr3-1,4}2)(η-Cp*)]2(μ-η1:η1-C2O2) Journal of the American Chemical Society, volume 130, issue 42, pages 13816–13817. doi:10.1021/ja8059792
